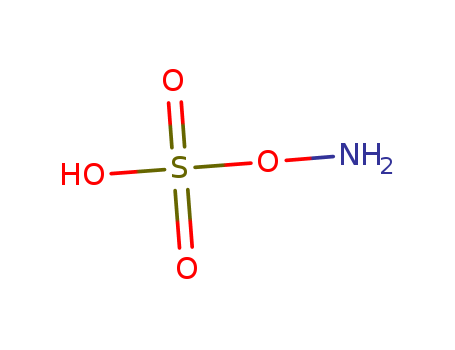


CasNo: 2950-43-8
MF: H3NO4S
Appearance: white to slightly beige crystalline powder
|
Purification Methods |
Stir the solid vigorously with anhydrous Et2O and filter it off using large volumes of dry Et2O. Drain dry at the pump for 5minutes and then for 12-14hours in a vacuum. Store it in a vacuum desiccator over conc H2SO4. Determine the purity by oxidation of iodide to I2. It must be stored in a dry atmosphere at 0-4o. It decomposes slowly in H2O at 25o and more rapidly above this temperature. [Matsuguma & Andrieth Inorg Synth V 122 1957.] |
|
General Description |
Hydroxylamine-O-sulfonic acid is a versatile synthetic reagent. It is widely employed as a nucleophile and as an electrophile in various organic syntheses. It can be synthesized by reacting hydroxylamine sulfate with 30% fuming H2SO4. |
InChI:InChI=1/H3NO4S/c1-5-6(2,3)4/h1H2,(H,2,3,4)
Multiple lines of evidence have indicate...
An approach to the synthesis of spirocyc...
A new method for the production of O-sub...
The imidazolium compounds of the formula...
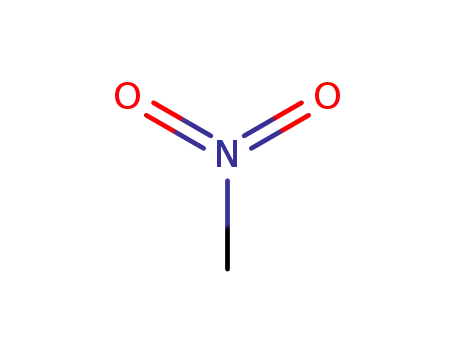
nitromethane

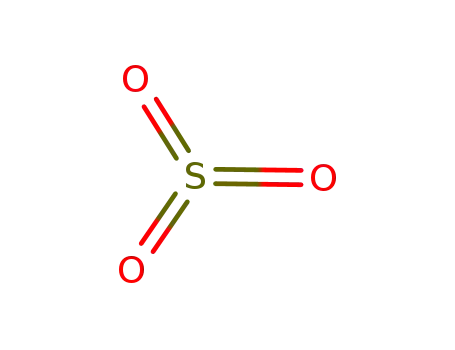
sulfur trioxide

carbon dioxide

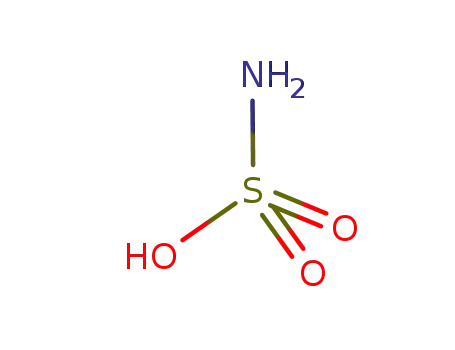
aminosulfonic acid

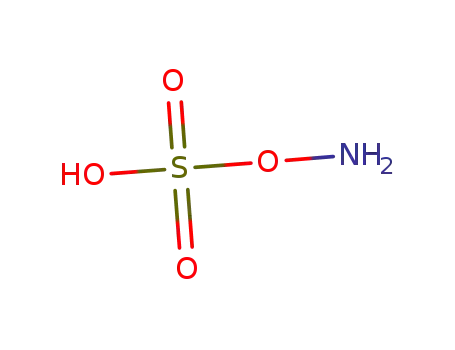
hydroxylamine-O-sulfonic acid
| Conditions | Yield |
|---|---|
|
above 0°C;
|
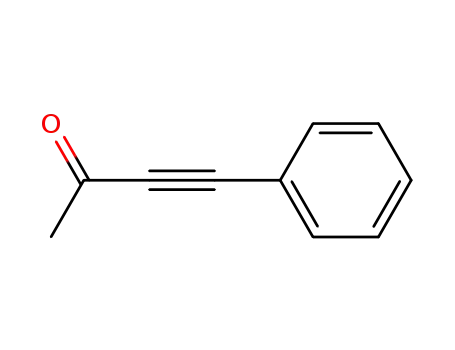
4-phenyl-3-butyne-2-one

pyridazin-1-ium-1-amine iodide

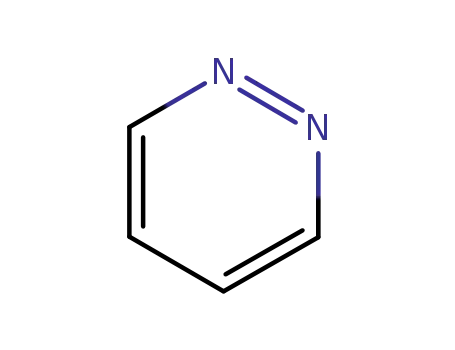
1,2-diazine


hydroxylamine-O-sulfonic acid
| Conditions | Yield |
|---|---|
|
With potassium hydrogencarbonate; potassium hydroxide; In water;
|
44% |

nitromethane

sulfur trioxide
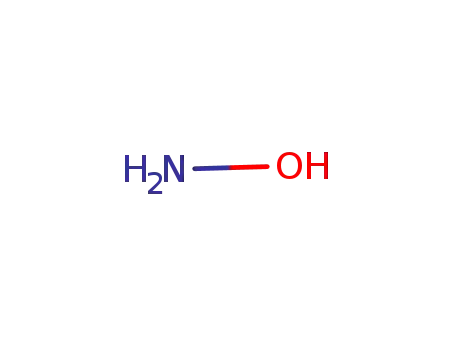
hydroxylamine
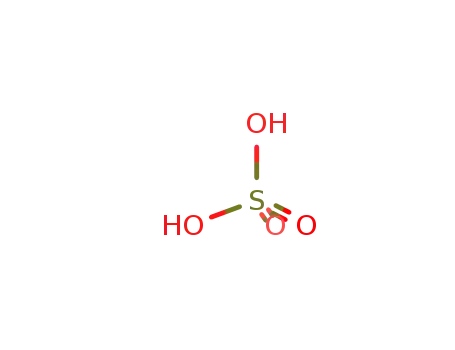
sulfuric acid
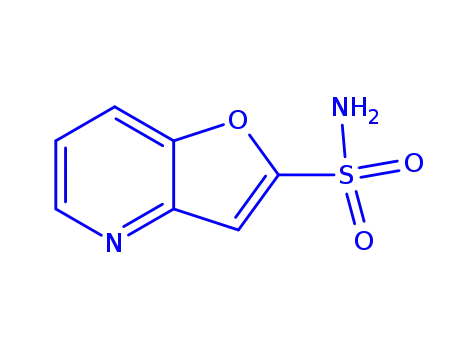
2-sulfamoylfuro[3,2-b]pyridine
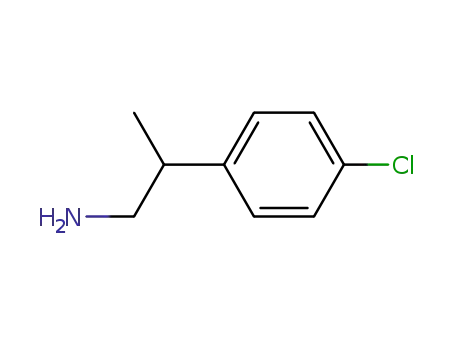
p-Chloro-b-methylphenylethylamine
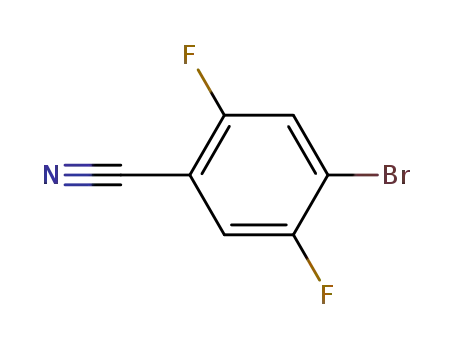
4-bromo-2,5-difluoro-benzonitrile
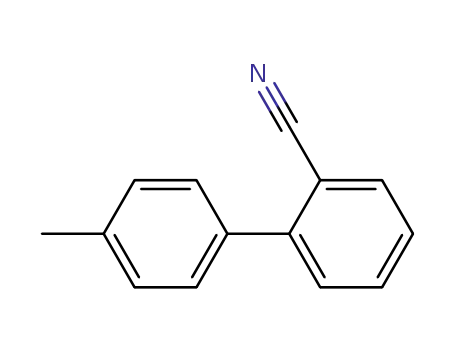
2-Cyano-4'-methylbiphenyl