


CasNo: 552-89-6
MF: C7H5NO3
Appearance: Yellow crystalline powder or needles
|
Preparation |
2-Nitrobenzaldehyde is synthesized from 2-Nitrotoluene by nitration and oxidation.Synthesis of 2-Nitrobenzaldehyde from 2-Nitrotoluene |
|
Synthesis Reference(s) |
Synthetic Communications, 19, p. 3385, 1989 DOI: 10.1080/00397918908052745 |
|
Purification Methods |
Crystallise the aldehyde from toluene (2-2.5mL/g) by addition of 7mL pet ether (b 40-60o) for 1mL of solution. It can also be distilled under reduced pressures. [Beilstein 7 IV 584.] |
|
Definition |
ChEBI: 2-nitrobenzaldehyde is benzaldehyde substituted at the ortho-position with a nitro group. It is a C-nitro compound and a member of benzaldehydes. |
|
Application |
2-Nitrobenzaldehyde can react with chitosan to form 2-nitrobenzyl-chitosan, which is soluble in trifluoroacetic acid and can also be electrospun into nanofiber matrices. On photolysis, the nitrobenzyl group is cleaved to form neat chitosan nanofiber matrices. The same principle has been applied for the preparation of gelatin nanofiber matrices. The condensation of o-NBA with 2-aminobenzothiazole forms o-nitrobenzylidene 2-aminobenzothiazole, a Schiff′s base that can react with metal ions to form complexes. |
|
General Description |
The 2-Nitrobenzyl group of 2-nitrobenzaldehyde is photolabile that can be cleaved when exposed to UV-light. |
InChI:InChI=1/C7H5NO3/c9-5-6-3-1-2-4-7(6)8(10)11/h1-5H
The first bimagnetic ionic liquid based ...
Nitrolysis of 2,6,8,12-tetraacetyl-4,10-...
The nitration of benzaldehyde can be car...
Oximes are oxidatively deprotected by th...
The barrier to rotation about the carbon...
A combination of iodic acid with ammoniu...
A highly efficient, selective and green ...
A variety of deactivated arenes were nit...
Malignant gliomas are the most common br...
Considering the outstanding characterist...
-
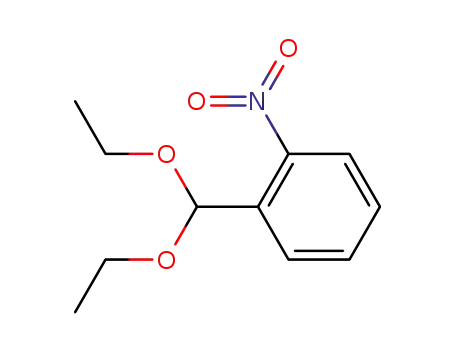
o-nitrobenzaldehyde diethylacetal

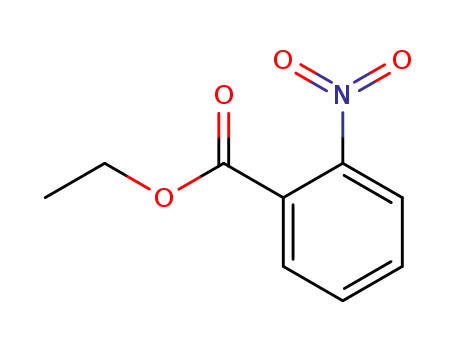
ethyl 2-nitrobenzoate

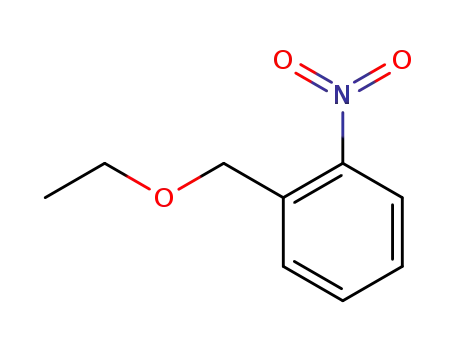
2-ethoxymethyl-1-nitrobenzene


2-nitro-benzaldehyde
| Conditions | Yield |
|---|---|
|
aluminum oxide; In gas; at 350 ℃; Yield given;
|
|
|
aluminum oxide; In gas; at 200 ℃; Yield given;
|
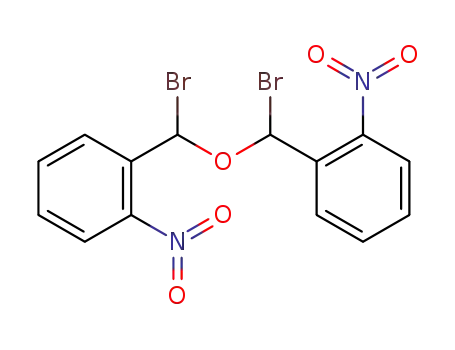
bis-(α-bromo-2-nitro-benzyl)-ether

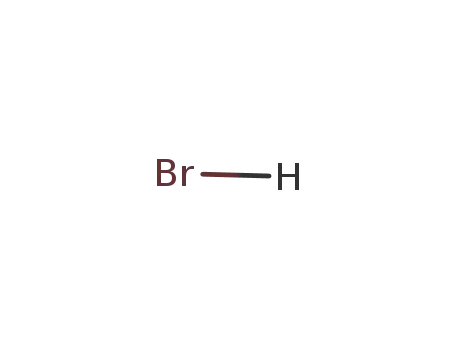
hydrogen bromide


2-nitro-benzaldehyde
| Conditions | Yield |
|---|---|
|
|
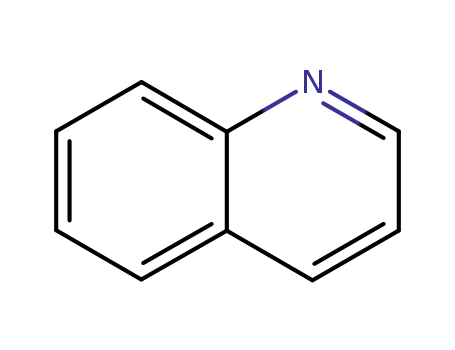
quinoline
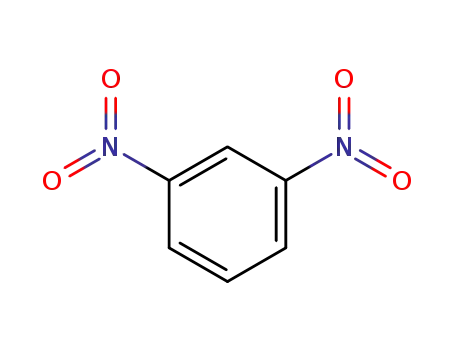
meta-dinitrobenzene
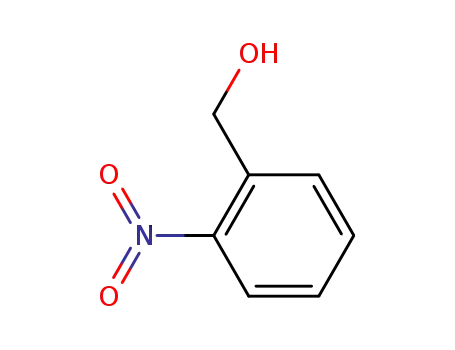
2-Nitrobenzyl alcohol
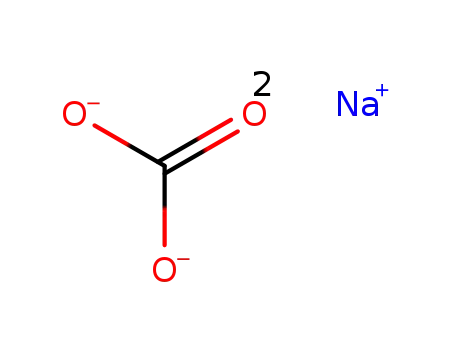
sodium carbonate
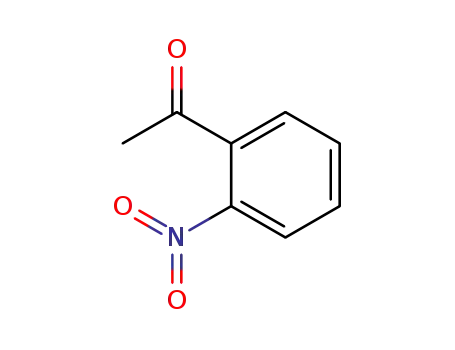
2-acetylnitrobenzene
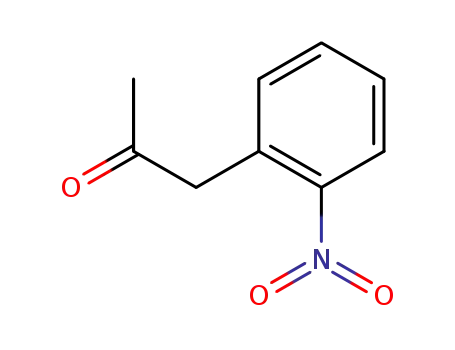
(2-nitrophenyl)acetone

(+/-)-2-(2-nitrophenyl)oxirane
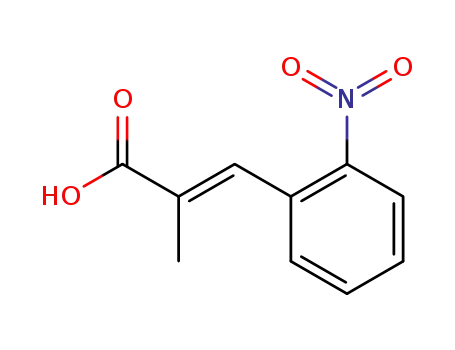
2-methyl-3t-(2-nitro-phenyl)-acrylic acid