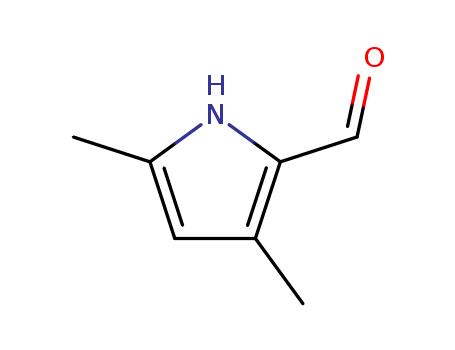
Factory Supply High Purity 2199-58-8 with Reasonable Price, Buy High Grade 3,5-Dimethyl-1H-pyrrole-2-carboxaldehyde
- Molecular Formula:C7H9NO
- Molecular Weight:123.155
- Appearance/Colour:yellow solid.
- Vapor Pressure:0.0452mmHg at 25°C
- Melting Point:95-99 °C(lit.)
- Refractive Index:1.576
- Boiling Point:237.3 °C at 760 mmHg
- PKA:16.23±0.50(Predicted)
- Flash Point:103.2 °C
- PSA:32.86000
- Density:1.099 g/cm3
- LogP:1.44400
3,5-Dimethylpyrrole-2-carboxaldehyde(Cas 2199-58-8) Usage
InChI:InChI=1/C7H9NO/c1-5-3-6(2)8-7(5)4-9/h3-4,8H,1-2H3
2199-58-8 Relevant articles
-
Murakami et al.
, p. 671,672 (1969)
-
Fluorogenic Ubiquinone Analogue for Monitoring Chemical and Biological Redox Processes
Greene, Lana E.,Godin, Robert,Cosa, Gonzalo
, p. 11327 - 11334 (2016)
We report herein the synthesis and chara...
Halogenated BODIPY photosensitizers: Photophysical processes for generation of excited triplet state, excited singlet state and singlet oxygen
Hu, Wenbin,Liu, Jiatian,Luo, Lin,Zhang, Rui,Zhang, Xian-Fu
, (2022/02/09)
We have systematically examined the form...
BOPHY-Fullerene C60 Dyad as a Photosensitizer for Antimicrobial Photodynamic Therapy
Gonzalez Lopez, Edwin J.,Sarotti, Ariel M.,Martínez, Sol R.,Macor, Lorena P.,Durantini, Javier E.,Renfige, Melisa,Gervaldo, Miguel A.,Otero, Luis A.,Durantini, Andrés M.,Durantini, Edgardo N.,Heredia, Daniel A.
, (2022/01/08)
A novel BOPHY–fullerene C60 dyad (BP-C60...
Synthesis and study of organoselenium compound: DNA/Protein interactions, in vitro antibacterial, antioxidant, anti-inflammatory activities and anticancer activity against carcinoma cells
Dhavan, Pratik P.,Jadhav, Bhaskar L.,Manjare, Sudesh T.,Shelar, Divyesh S.,Singh, Pinky R.,Vaidya, Shashikant P.
, (2021/07/25)
New organoselenium compound was synthesi...
Virtual Screening Identifies Irreversible FMS-like Tyrosine Kinase 3 Inhibitors with Activity toward Resistance-Conferring Mutations
Bensinger, Dennis,Stubba, Daniel,Cremer, Anjali,Kohl, Vanessa,Wa?mer, Theresa,Stuckert, Johanna,Engemann, Victoria,Stegmaier, Kimberly,Schmitz, Katja,Schmidt, Boris
supporting information, p. 2428 - 2446 (2019/03/11)
The use of covalent irreversible binding...
2199-58-8 Process route
-
- 2199-58-8
3,5-dimethylpyrrole-2-carbaldehyde
-
- 10087-64-6
2,2'-bipyrrole
-
- 625-82-1
2,4-dimethyl-1H-pyrrole
-
- 2199-58-8
3,5-dimethylpyrrole-2-carbaldehyde
Conditions
| Conditions |
Yield |
|
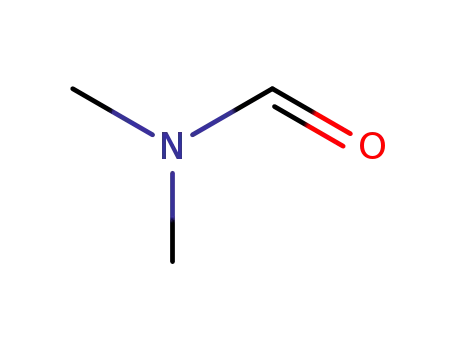
N,N-dimethyl-formamide; With trichlorophosphate; at 0 ℃; for 0.333333h;
2,4-dimethyl-1H-pyrrole; In 1,2-dichloro-ethane; at 0 ℃; for 0.5h; Heating / reflux;
With sodium acetate; In 1,2-dichloro-ethane; for 0.333333h; Heating / reflux;
|
80% |
|
Multi-step reaction with 3 steps
1: ethyl magnesium bromide; diethyl ether
2: aqueous KOH
With potassium hydroxide; diethyl ether; ethylmagnesium bromide;
|
|
2199-58-8 Upstream products
-
625-82-1
2,4-dimethyl-1H-pyrrole
-
74-90-8
hydrogen cyanide
-
77287-34-4
formamide
-
68-12-2
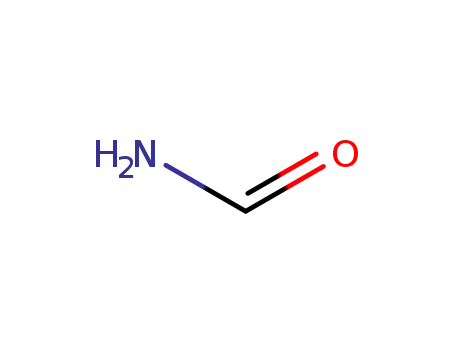
N,N-dimethyl-formamide
2199-58-8 Downstream products
-
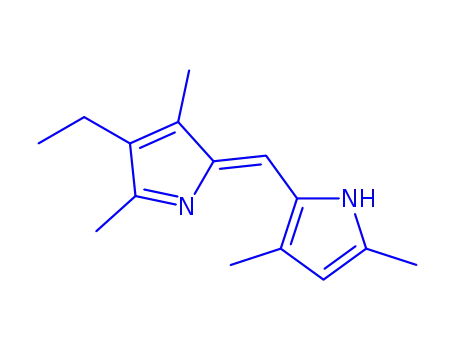
801999-64-4
2,2'-(3,3',5,5'-Tetramethyl-4-ethyl)-dipyrrylmethen
-
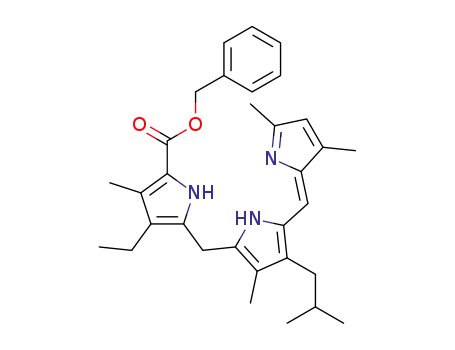
775549-69-4
5-{5-[3,5-Dimethyl-pyrrol-(2Z)-ylidenemethyl]-4-isobutyl-3-methyl-1H-pyrrol-2-ylmethyl}-4-ethyl-3-methyl-1H-pyrrole-2-carboxylic acid benzyl ester
-
194413-58-6
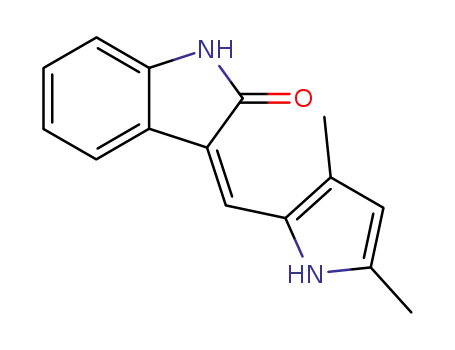
semaxanib
-
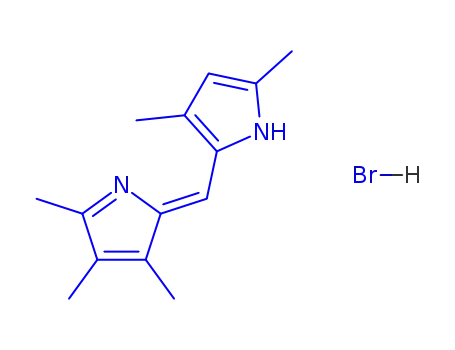
293733-87-6
2,2',3,3',4',5,5'-pentamethyldipyrrolylmethene-2,2' hydrobromide