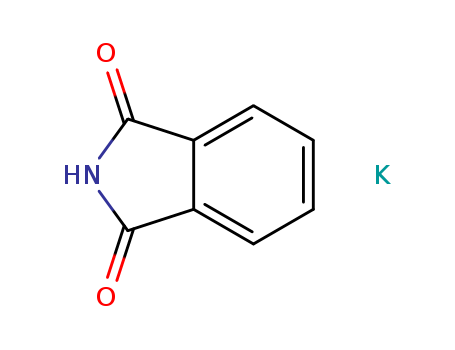


CasNo: 1074-82-4
MF: C8H4KNO2
Appearance: greenish white powder
|
Purification Methods |
The solid may contain phthalimide and K2CO3 from hydrolysis. If too much hydrolysis has occurred (this can be checked by extraction with cold Me2CO in which the salt is insoluble, evaporate the Me2CO and weigh the residue), it would be better to prepare it afresh. If little hydrolysis has occurred, then recrystallise it from a large volume of EtOH, and wash the solid with a little Me2CO and dry it in a continuous vacuum to constant weight. [Salzerg & Supriawski Org Synth Coll Vol I 119 1941, Raman & IR: Hase J Mol Struct 48 33 1978, Dykman Chem Ind (London) 40 1972, IR, NMR: Assef et al. Bull Soc Chim Fr II 167 1979, Beilstein 21/10 V 270.] |
InChI:InChI=1/C8H5NO2.K/c10-7-5-3-1-2-4-6(5)8(11)9-7;/h1-4H,(H,9,10,11);/q;+1
Two aliphatic ether Schiff base lanthani...
Aims: Partial PPARγ agonists attracted s...
Coumarin skelton holds substantial promi...
A straightforward synthesis of triphenyl...
Eutectics are emerging as promising cand...
A straight forward synthesis and efficie...
Three series of bitopic benzopyranomorph...
A method for synthesizing 2-(2-aminoetho...
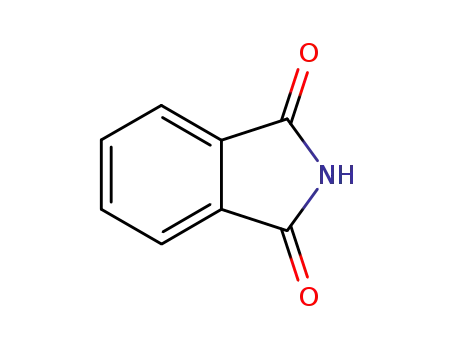
phthalimide

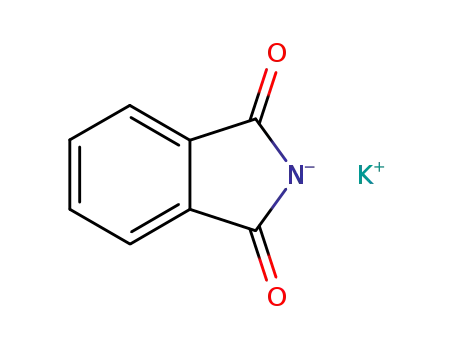
potassium phtalimide
| Conditions | Yield |
|---|---|
|
With potassium methanolate; In ethanol; water; at 60 - 65 ℃; for 13h; Reagent/catalyst; Solvent; Temperature;
|
98% |
|
With potassium hydroxide; In ethanol; at 75 ℃; for 1h;
|
96% |
|
With acrylonitrile; In methanol; ethanol; at 20 ℃; for 3h;
|
95% |
|
With potassium hydroxide; In ethanol; for 2h;
|
91.5% |
|
With potassium hydroxide; In ethanol; at 60 ℃; for 24h;
|
91% |
|
With potassium hydroxide; In ethanol; for 2h; Reflux;
|
90.3% |
|
With potassium hydroxide; In ethanol; for 2h; Reflux;
|
90.3% |
|
With potassium hydroxide; In methanol; ethanol; for 1h; Inert atmosphere;
|
89% |
|
With potassium hydroxide; In methanol; water; at 20 ℃;
|
82% |
|
With potassium hydroxide; In ethanol; water; Reflux;
|
78% |
|
With potassium hydroxide; In ethanol; water; at 0 ℃; Reflux;
|
71% |
|
With potassium hydroxide; In methanol; water; for 72h; Heating;
|
57% |
|
With potassium hydroxide; In ethanol; water; Reflux;
|
30% |
|
With potassium hydroxide; In ethanol; water; Reflux;
|
|
|
With KOH; In ethanol;
|
|
|
With potassium hydroxide; In ethanol;
|
|
|
With potassium hydroxide; In ethanol; Reflux;
|
|
|
With potassium hydroxide; In ethanol;
|
|
|
With potassium hydroxide; In methanol; ethanol; at 20 ℃; for 3h;
|
|
|
With ethanol; potassium hydroxide; for 7h; Reflux;
|
|
|
With potassium hydroxide; In ethanol; Microbiological reaction;
|
12 g |
|
With potassium hydroxide; In acetone;
|
|
|
With potassium hydroxide; In ethanol; at 55 ℃; for 3h;
|
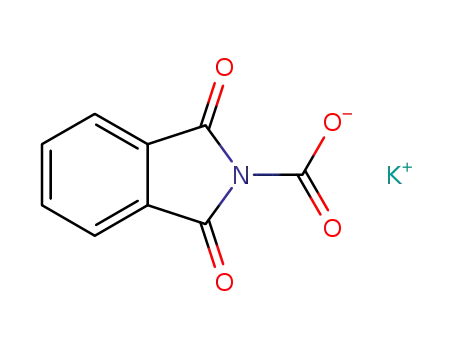
C9H4NO4(1-)*K(1+)

carbon dioxide


potassium phtalimide
| Conditions | Yield |
|---|---|
|
With nitrogen; at 65 ℃; for 0.666667h;
|
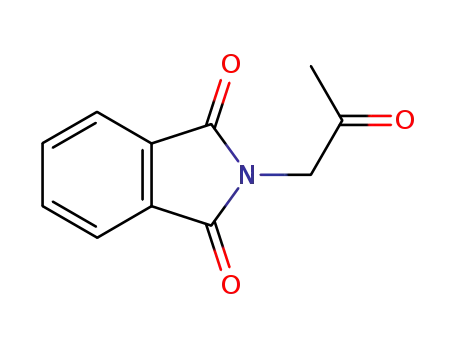
N-acetonylphthalimide

phthalimide

C9H4NO4(1-)*K(1+)
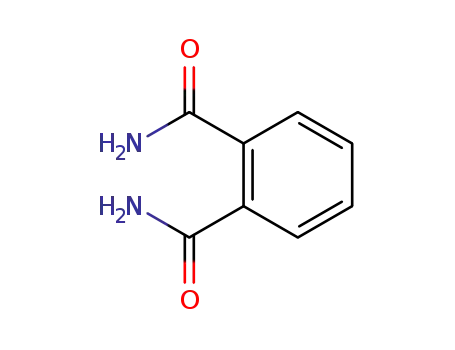
phthalamide
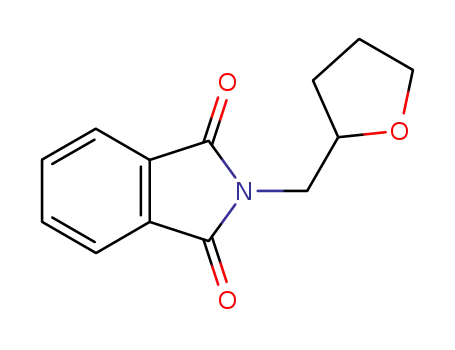
2-((tetrahydrofuran-2-yl)methyl)isoindoline-1,3-dione
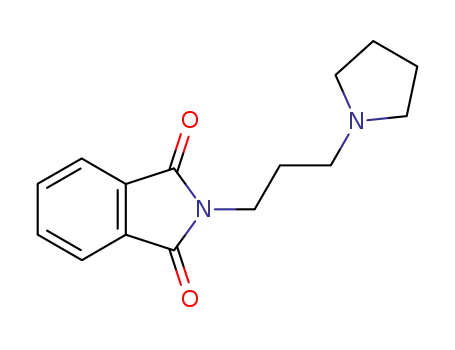
2-(3-pyrrolidinylpropyl)-1H-isoindole-1,3(2H)-dione
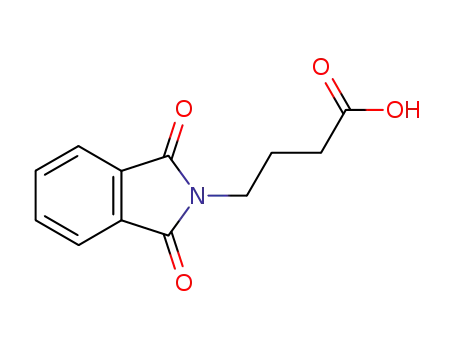
4-phthalimidobutyric acid
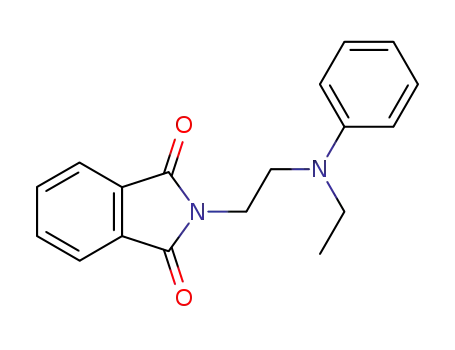
N-[2-(N-ethyl-anilino)-ethyl]-phthalimide