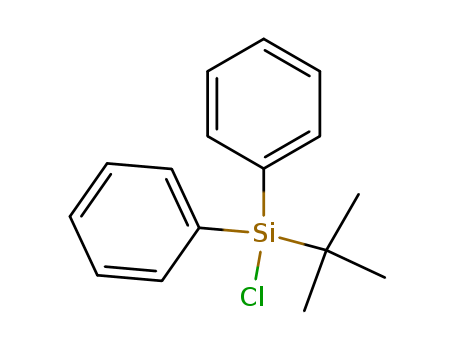


CasNo: 58479-61-1
MF: C16H19ClSi
Appearance: Colorless-light yellow to brown liquid
|
Preparation |
a dry 1 L, three-necked round bottomed flask is equipped with a magnetic stirring bar, a 500mL equalizing dropping funnel fitted with a rubber septum, a reflux condenser, and nitrogen inlet tube. The flask is flushed with nitrogen, then charged with 127 g (0.5 mol) of diphenyldichlorosilane in 300mL of redistilled pentane. A solution of tbutyllithium in pentane (500 mL, 0.55 mol), is transferred under nitrogen pressure to the dropping funnel using a stainless steel, double-tip transfer needle. This solution is slowly added to the contents of the flask and when the addition is complete, the mixture is refluxed 30 h under nitrogen with stirring. The suspension is allowed to cool to rt, the precipitated lithium chloride is rapidly filtered through a pad of Celite, and the latter is washed with 200mL of pentane. The solvent is removed by evaporation, and the colorless residue is distilled through a short (10 cm), Vigreux column, to give 125–132 g of the colorless title compound. |
|
Purification Methods |
Purify it by repeated fractional distillaton. It is soluble in DMF and pentane [Hanessian & Lavalee Can J Chem 53 2975 1975, Robl et al. J Med Chem 34 2804 1991]. [Beilstein 4 IV 4076 for tert-butylchlorodimethylsilane.] |
|
Physical properties |
colorless liquid, bp 93–95°C/0.015 mmHg; n20 D 1.5680; d 1.057 g cm?3. |
InChI:InChI=1/C16H19ClSi/c1-16(2,3)18(17,14-10-6-4-7-11-14)15-12-8-5-9-13-15/h4-13H,1-3H3
-
We present the preparation of tert-butyl...
PROBLEM TO BE SOLVED: To provide a metho...
Chlorosilanes are versatile reagents in ...
A simple and highly efficient iron-catal...
tertiary butyl chloride

diphenylsilyl dichloride

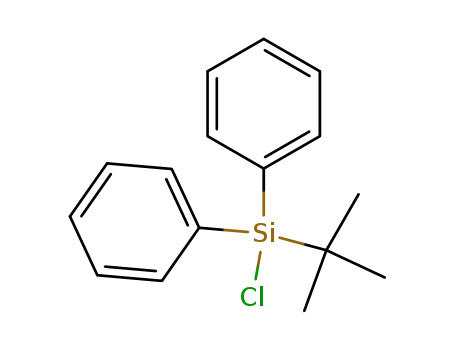
tert-butylchlorodiphenylsilane
| Conditions | Yield |
|---|---|
|
tertiary butyl chloride; With magnesium; In tetrahydrofuran; at 60 ℃; for 2h;
With lithium chloride; copper(I) bromide; In tetrahydrofuran; at 20 ℃; for 1h;
diphenylsilyl dichloride; In tetrahydrofuran; at 50 ℃; for 6h; Reagent/catalyst; Temperature;
|
87% |
|
With copper(l) cyanide; magnesium; In tetrahydrofuran; for 5h; Heating;
|
80% |
|
With magnesium; In tetrahydrofuran; hexane;
|
75% |

diphenylsilyl dichloride


tert-butylchlorodiphenylsilane
| Conditions | Yield |
|---|---|
|
copper (II) acetylacetonate;
|
85% |

t-butyl diphenylsilanol
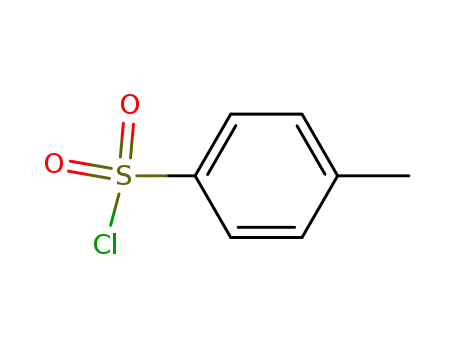
p-toluenesulfonyl chloride
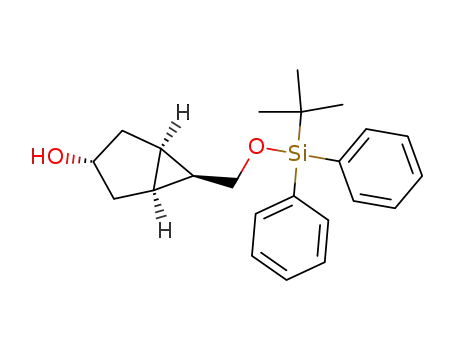
(1S,3S,5R,6S)-6-(tert-Butyl-diphenyl-silanyloxymethyl)-bicyclo[3.1.0]hexan-3-ol

tertiary butyl chloride
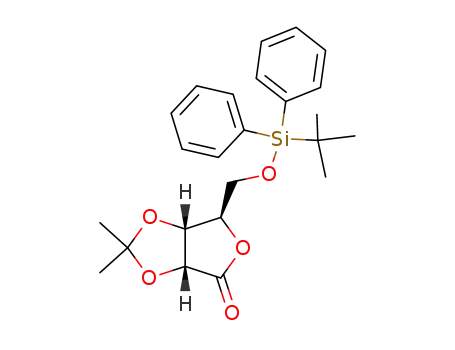
5-(tert-butyldiphenylsilyl)-2,3-O-isopropylidene-D-ribono-1,4-lactone
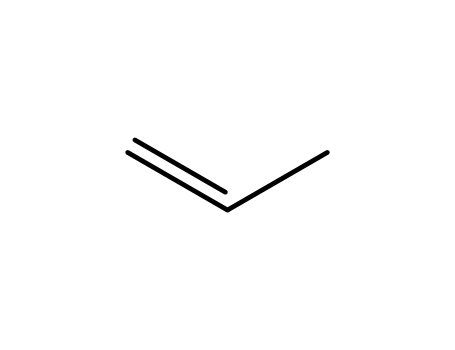
propene
tert-butyl(pent-4-en-1-yloxy)diphenylsilane

tert-Butyl-diphenyl-vinyloxy-silane