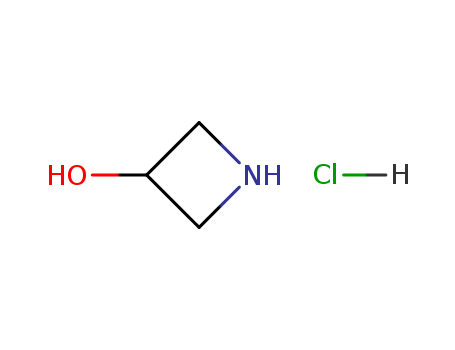


CasNo: 18621-18-6
MF: C3H7NO.HCl
Appearance: white crystal
InChI:InChI=1/C3H7NO.ClH/c5-3-1-4-2-3;/h3-5H,1-2H2;1H
A thoroughly optimized and robust proces...
1-Azabicyclo[1.1.0]butane 2 was successf...
The invention discloses a synthetic meth...
The invention discloses a 3-nitrile meth...
The invention discloses an aryl removing...
The invention discloses a method for pre...
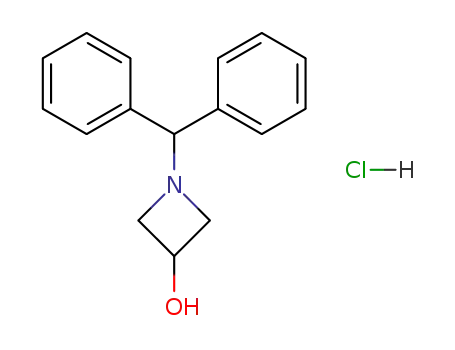
1-(diphenylmethyl)-3-azetidinol hydrochloride

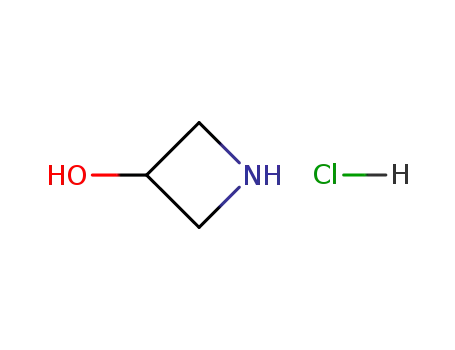
azetion-3-ol hydrochloride
| Conditions | Yield |
|---|---|
|
With hydrogen; palladium on activated charcoal; In ethanol; water; at 20 ℃; for 4h; under 2625.21 Torr;
|
100% |
|
With palladium hydroxide on carbon; hydrogen; In ethanol; at 20 ℃; for 12h; under 3040.2 Torr; Inert atmosphere;
|
94% |
|
With hydrogen; palladium hydroxide on carbon; In ethanol; at 20 ℃; for 12h; under 3040.2 Torr;
|
94% |
|
With hydrogen; palladium(II) hydroxide/carbon; In methanol; for 48h; under 2844.39 Torr;
|
52% |
|
With hydrogen; palladium hydroxide on carbon; In methanol; for 48h; under 2844.39 Torr;
|
52% |
|
With hydrogenchloride; hydrogen; palladium on activated charcoal;
|
|
|
With hydrogen; palladium dihydroxide; In methanol; for 72h; under 2844.3 Torr;
|
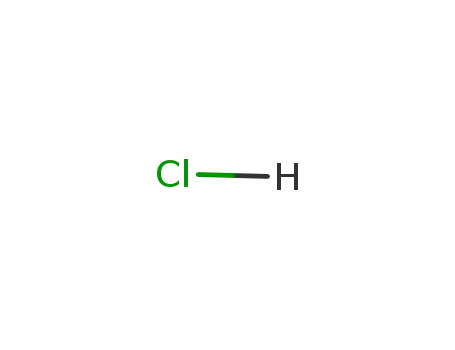
hydrogenchloride

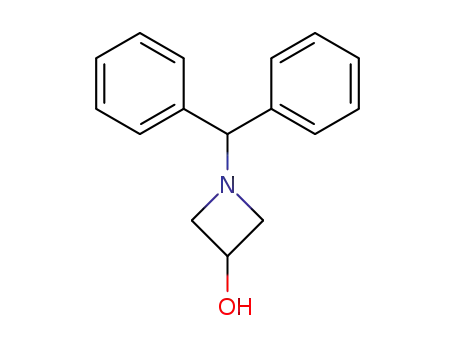
1-(diphenylmethyl)-3-hydroxyazetidine


azetion-3-ol hydrochloride
| Conditions | Yield |
|---|---|
|
With hydrogen; 10% palladium on activated carbon; In 1,4-dioxane; methanol; at 60 ℃; for 6h; under 2068.65 Torr;
|
68% |

1-(diphenylmethyl)-3-azetidinol hydrochloride
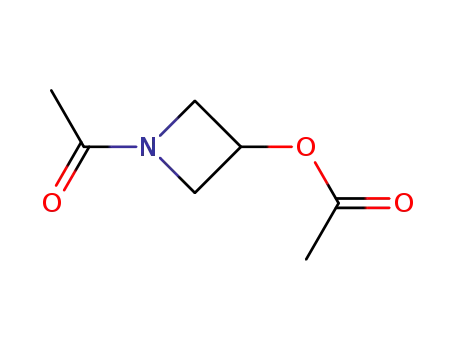
N-acetyl-3-acetoxyazetidine

1-azabicyclo<1.1.0>butane

hydrogenchloride
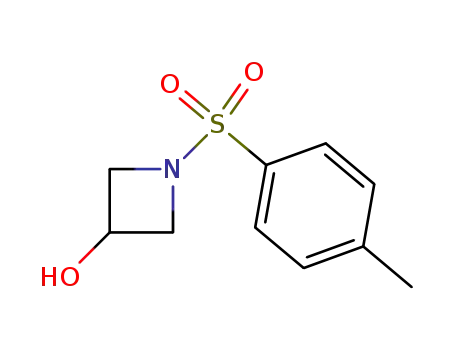
1-(p-Toluenesulfonyl)azetidin-3-ol
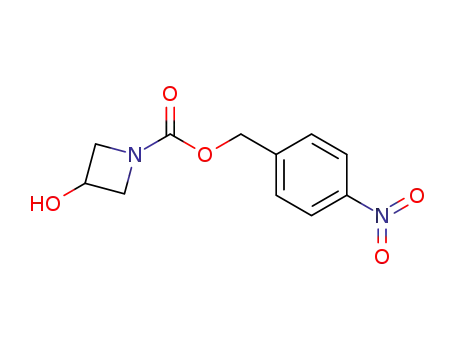
3-hydroxy-1-(p-nitrobenzyloxycarbonyl)azetidine
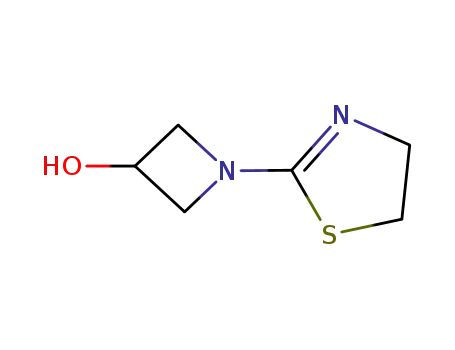
1-(1,3-thiazolin-2-yl-)azetidin-3-ol
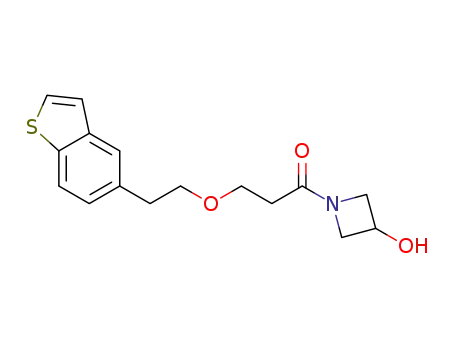
1-(3-(2-(1-benzothiophen-5-yl)ethoxy)propionyl)azetidin-3-ol