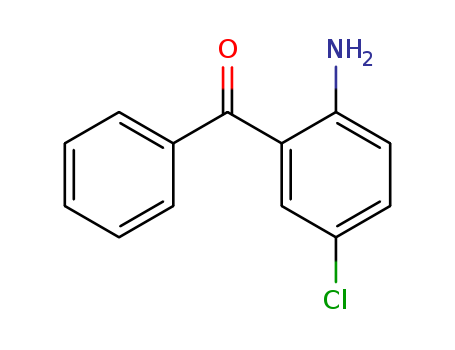


CasNo: 719-59-5
MF: C13H10ClNO
Appearance: yellow powder
|
Synthesis Reference(s) |
Tetrahedron Letters, 35, p. 6811, 1994 DOI: 10.1016/0040-4039(94)85011-9 |
|
General Description |
2-Amino-5-chlorobenzophenone is a organic nonlinear optical material. |
InChI:InChI=1/C13H10ClNO/c14-10-6-7-12(15)11(8-10)13(16)9-4-2-1-3-5-9/h1-8H,15H2
The present invention proposes a method ...
Herein, an iridium(iii)-catalysed electr...
We describe a novel one-pot three-step r...
An efficient copper-catalyzed electrophi...
![(R)-7-Chloro-3-((S)-hydroxy-phenyl-methyl)-1-methyl-5-phenyl-1,3-dihydro-benzo[e][1,4]diazepin-2-one](/upload/2024/7/f38ca239-5629-4631-880c-67baac00f8cb.png)
(R)-7-Chloro-3-((S)-hydroxy-phenyl-methyl)-1-methyl-5-phenyl-1,3-dihydro-benzo[e][1,4]diazepin-2-one

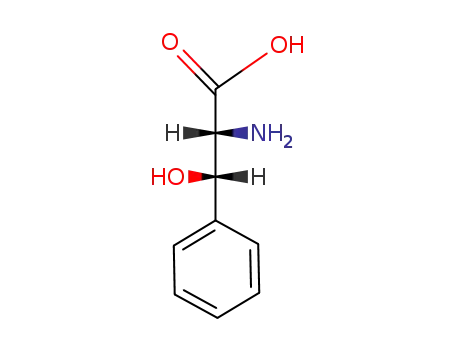
dl-threo-phenylserine

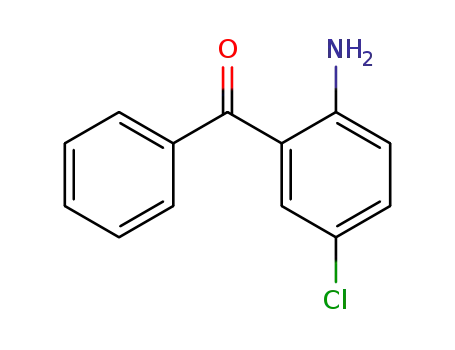
5-chloro-2-aminobenzophenone
| Conditions | Yield |
|---|---|
|
With hydrogenchloride; In acetic acid; for 16h; Heating;
|
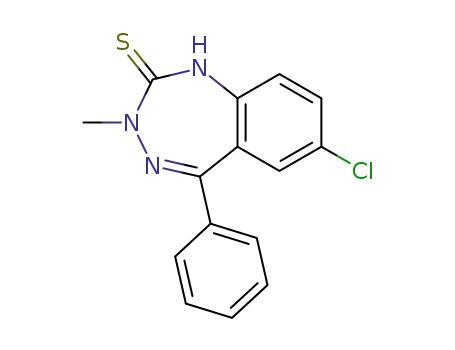
7-Chlor-3-methyl-5-phenyl-2-thioxo-3H-1,2-dihydro-1,3,4-benzotriazepins


5-chloro-2-aminobenzophenone

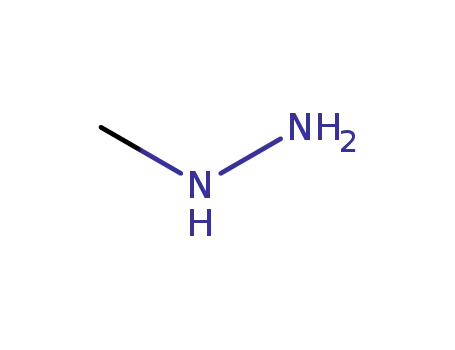
methylhydrazine
| Conditions | Yield |
|---|---|
|
With hydrogenchloride; for 10h; Heating;
|
98 % Spectr. |
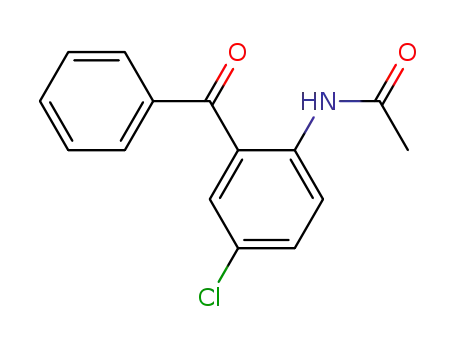
N-(2-benzoyl-4-chlorophenyl)acetamide
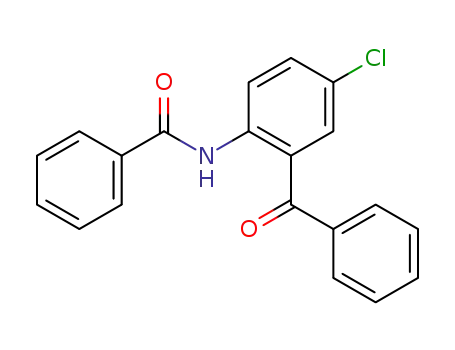
N-Benzoyl-2-amino-5-chlorobenzophenone
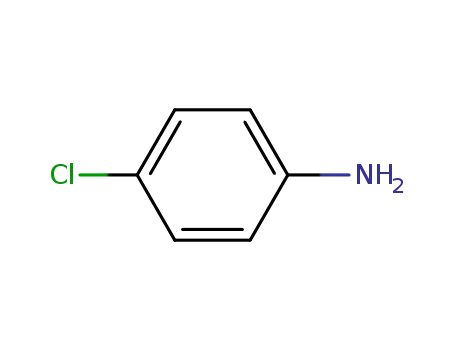
4-chloro-aniline
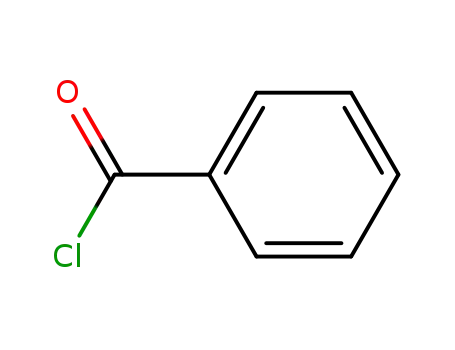
benzoyl chloride
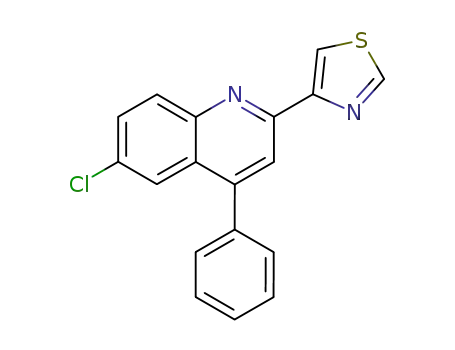
6-chloro-4-phenyl-2-thiazol-4-yl-quinoline
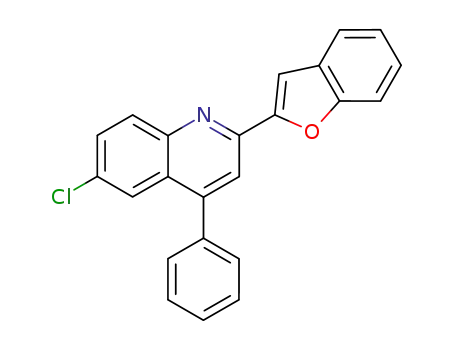
2-benzofuran-2-yl-6-chloro-4-phenyl-quinoline
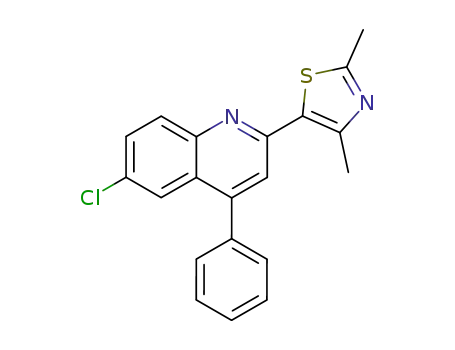
6-chloro-2-(2,4-dimethyl-thiazol-5-yl)-4-phenyl-quinoline
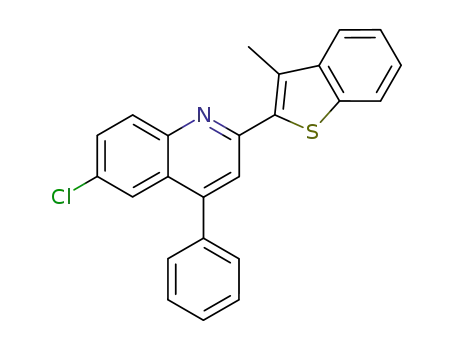
6-chloro-2-(3-methyl-benzo[b]thiophen-2-yl)-4-phenyl-quinoline