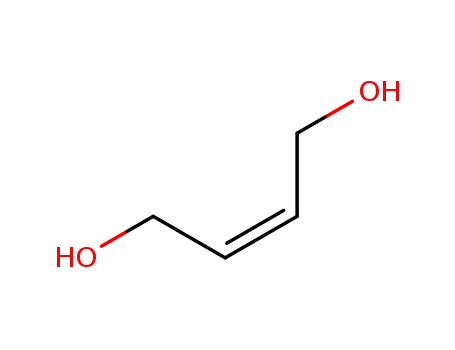


CasNo: 6117-80-2
MF: C4H8O2
Appearance: clear liquid
InChI:InChI=1/C4H8O2/c5-3-1-2-4-6/h1-2,5-6H,3-4H2/b2-1-
A catalyst consisting of platinum nanopa...
Cubic Pd nanoparticles were rapidly enca...
Internal alkynes were hydrogenated quant...
The hyperpolarization of nuclear spins i...
Soluble small molecular/polymeric ligand...
In this study,in situformed ruthenium hy...
The first continuous flow Z-selective ol...
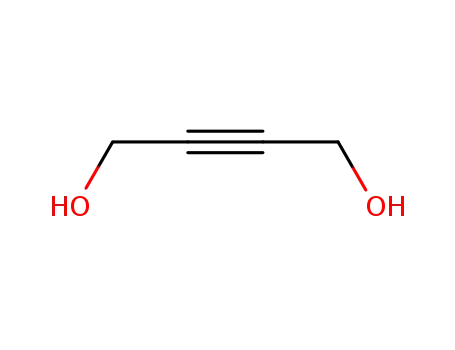
1,4-dihydroxybut-2-yne

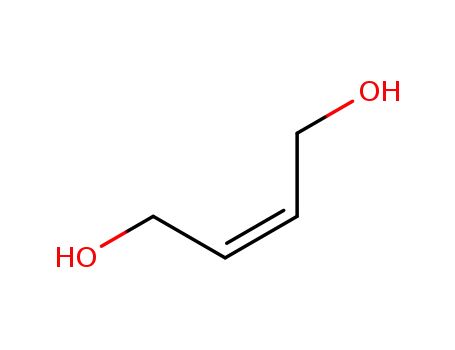
1,4-butenediol
| Conditions | Yield |
|---|---|
|
With quinoline; hydrogen; Lindlar's catalyst; In methanol; at 0 ℃;
|
100% |
|
With borane-ammonia complex; Cu2O; In ethanol; at 50 ℃; for 0.75h; stereoselective reaction; Sealed tube; Green chemistry;
|
100% |
|
With hydrogen; copper-palladium; silica gel; In ethanol; at 25 ℃; under 760.051 Torr;
|
99% |
|
With piperazine; hydrogen; In ethanol; at 80 ℃; for 24h; under 4500.45 Torr; diastereoselective reaction;
|
99% |
|
With hydrogen; montmorillonit-bipyridinpalladium(II)-acetate; In tetrahydrofuran; Ambient temperature;
|
98% |
|
With quinoline; hydrogen; Lindlar's catalyst; In various solvent(s); at 20 ℃; for 2h; under 760.051 Torr;
|
98% |
|
With hydrogen; In methanol; at 20 ℃; for 5.5h; under 760.051 Torr; Green chemistry;
|
98% |
|
With hydrogen; In methanol; for 5.5h; under 760.051 Torr; Time;
|
97% |
|
With borohydride exchange resin; hydrogen; nickel diacetate; In methanol; at 0 ℃; for 1h; under 760 Torr;
|
94% |
|
With hydrogen; In ethanol; at 100 ℃; for 24h; under 7500.75 Torr; chemoselective reaction;
|
94% |
|
With hydrogen; palladium amalgam; silica gel; In ethyl acetate; for 16h; under 760 Torr; Ambient temperature;
|
92% |
|
With pyridine; hydrogen; In ethyl acetate; at 25 ℃;
|
90% |
|
With hydrogen; quinoline; In ethanol; at 25 ℃; under 760 Torr;
|
80% |
|
With copper; zinc; In methanol; for 2h; Heating;
|
57% |
|
With methanol; nickel; at 25 - 50 ℃; Hydrogenation;
|
|
|
With methanol; palladium on activated charcoal; Hydrogenation;
|
|
|
With water; iron; at 50 ℃; Hydrogenation.unter Druck;
|
|
|
With copper; Hydrogenation;
|
|
|
With nickel; Hydrogenation;
|
|
|
With cobalt; Hydrogenation;
|
|
|
With platinum; Hydrogenation;
|
|
|
With zinc copper; ethanol;
|
|
|
With potassium hydroxide; Reduktion an einer versilberten Kupfer-Kathode;
|
|
|
With ethanol; nickel; at 25 - 50 ℃; Hydrogenation;
|
|
|
With hydrogen; ruthenium palladium;
|
|
|
With hydrogen; In water; at 50 ℃; under 15001.5 Torr; Reagent/catalyst; Temperature; Pressure; chemoselective reaction; Catalytic behavior; Kinetics;
|
|
|
With hydrogen; In ethanol; at 20 ℃; under 750.075 Torr; chemoselective reaction;
|
|
|
With hydrogen; In water; at 120 ℃; for 6h; under 15001.5 Torr; Pressure; Temperature; Time;
|
|
|
With hydrogen; In methanol; at 20 ℃; for 8h; Reagent/catalyst; Flow reactor;
|
|
|
With [1,4-bis(diphenylphosphino)butane](1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; para-hydrogen; In [(2)H6]acetone; for 0.0166667h; under 760.051 Torr; Solvent; Mechanism;
|

1,4-dihydroxybut-2-yne

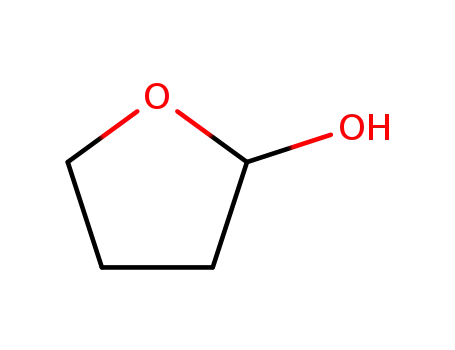
2-hydroxytetrahydrofuran

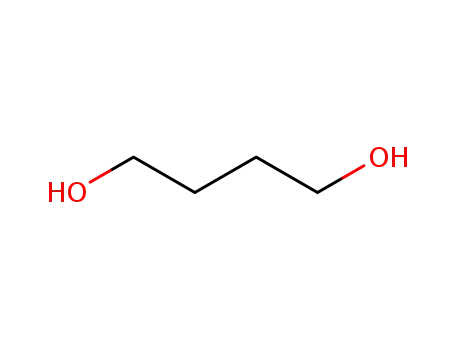
Butane-1,4-diol


1,4-butenediol
| Conditions | Yield |
|---|---|
|
With hydrogen; Montmorillonite-Ph2PPd(II); In tetrahydrofuran; at 25 ℃; for 2h; under 760 Torr; Product distribution; other catalysts (5percent Pd-C, Lindlar catalyst), other solvents (ethyl acetate);
|

1,4-dihydroxybut-2-yne
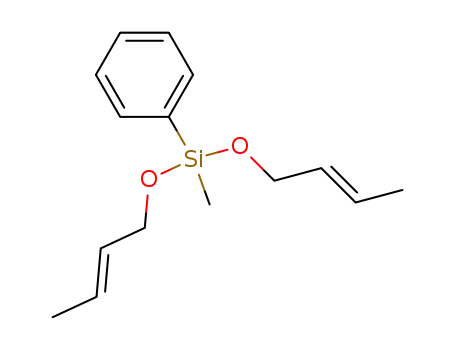
bis(2-butenoxy)methylphenylsilane
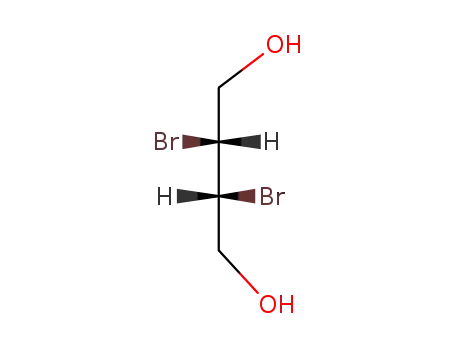
dl-2,3-dibromo-1,4-butanediol
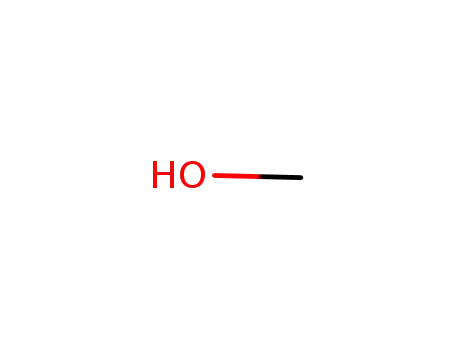
methanol

cis-1,4-dibromo-2-butene

1,3-dioxa-5-cycloheptene
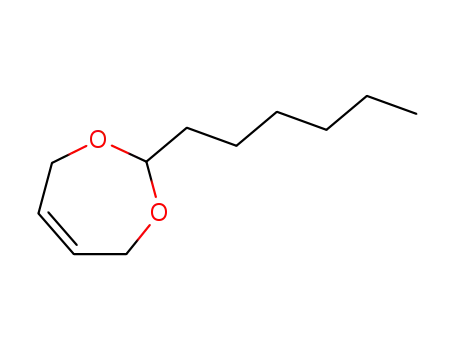
2-Hexyl-4,7-dihydro-[1,3]dioxepin
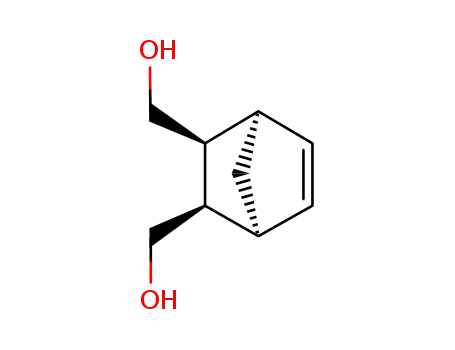
(1R,2S,3R,4S)-bicyclo[2.2.1]hept-5-ene-2,3-dimethanol