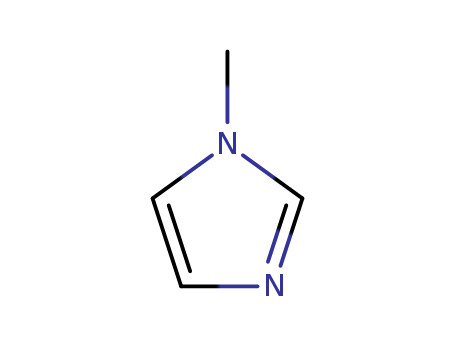


CasNo: 616-47-7
MF: C4H6N2
Appearance: colorless transparent liquid
|
Preparation |
1-Methylimidazole is prepared mainly by two routes industrially. The main one is acid-catalysed methylation of imidazole by methanol. The second method involves the Radziszewski reaction from glyoxal, formaldehyde, and a mixture of ammonia and methylamine. (CHO)2 + CH2O + CH3NH2 + NH3 → H2C2N(NCH3)CH + 3 H2O The compound can be synthesized on a laboratory scale by methylation of imidazole at the pyridine-like nitrogen and subsequent deprotonation. Similarly, 1-methylimidazole may be synthesized by first deprotonating imidazole to form a sodium salt followed by methylation. H2C2N(NH)CH + CH3I → [H2C2(NH)(NCH3)CH]I [H2C2(NH)(NCH3)CH]I + NaOH → H2C2N(NCH3)CH + H2O + NaI |
|
Flammability and Explosibility |
Notclassified |
|
Purification Methods |
Dry it with sodium metal and then distil it. Store it at 0o under dry argon. The picrate has m 159.5-160.5o (from H2O). [Beilstein 23 III/IV 568.] |
|
Definition |
ChEBI: 1-methyl-1H-imidazole is a 1H-imidazole having a methyl substituent at the N-1 position. It is a metabolite of 1-methyl-2-thioimidazole (methimazole). It inhibits bone resorption. |
|
General Description |
This product has been enhanced for catalysis. 1-Methylimidazole is a derivative of imidazole that is utilized in the manufacture of such classes of items as pharmaceuticals, pesticides, ion-exchange resins, dye intermediates, textile auxiliaries, photographic chemicals, and corrosion inhibitors. It is also used as a catalyst for manufacturing polyurethanes and a curing agent for epoxy resins. |
InChI:InChI=1/C4H6N2/c1-6-3-2-5-4-6/h2-4H,1H3
Vapor-phase N-alkylation syntheses of im...
Ionic liquids are of great importance fo...
This article describes the ionic and sup...
Temperature-induced change in reactivity...
Abstract: The reactions of imidazoles wi...
Photoredox catalysis has recently emerge...
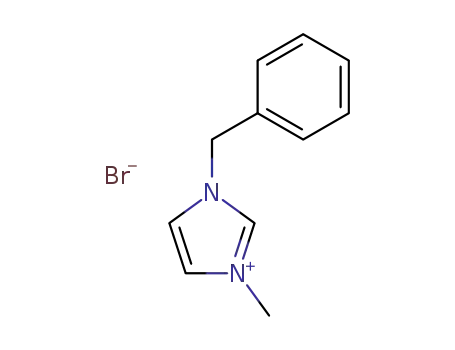
3-benzyl-1-methylimidazolium bromide

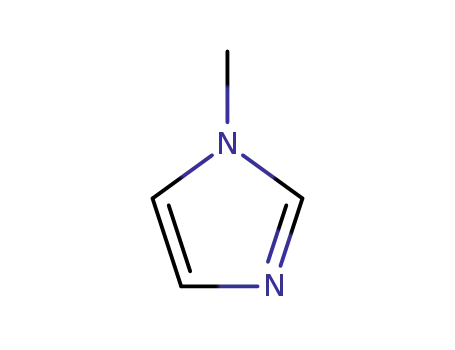
1-methyl-1H-imidazole

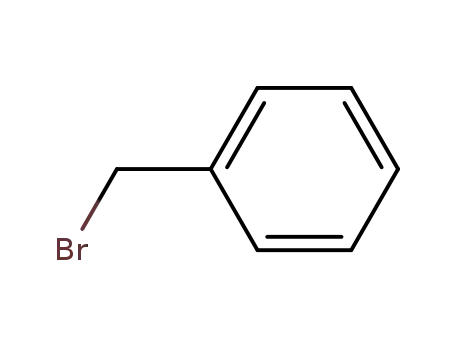
benzyl bromide
| Conditions | Yield |
|---|---|
|
at 260 - 300 ℃; under 12 Torr;
|

1-(p-methoxybenzoyl)-3-methylimidazolium chloride


1-methyl-1H-imidazole


4-methoxy-benzoyl chloride
| Conditions | Yield |
|---|---|
|
In dichloromethane; at 25 ℃; Rate constant; Equilibrium constant;
|
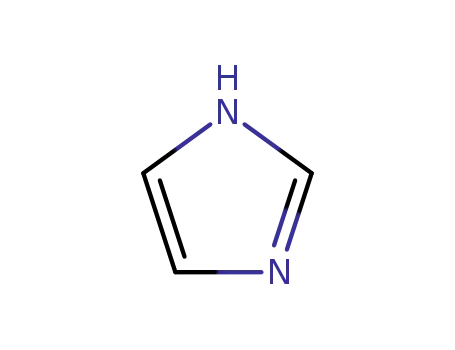
1H-imidazole
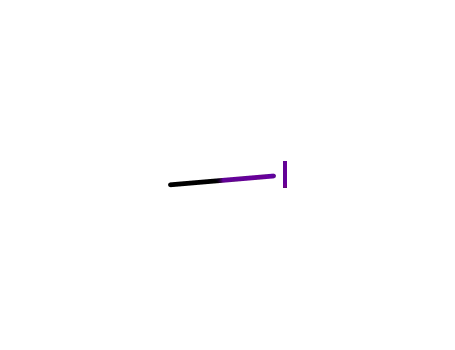
methyl iodide
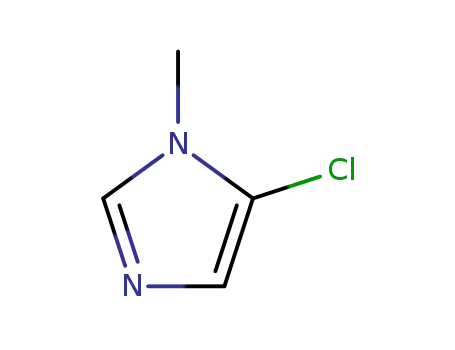
N-methyl-5-chloroimidazole
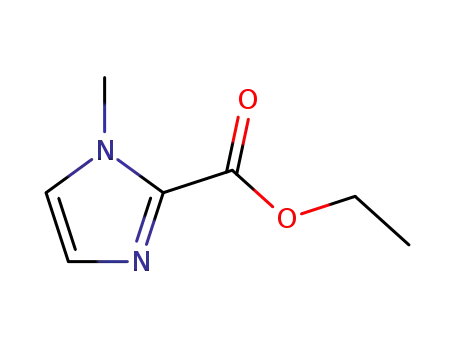
ethyl 1-methyl-1H-imidazole-2-carboxylate
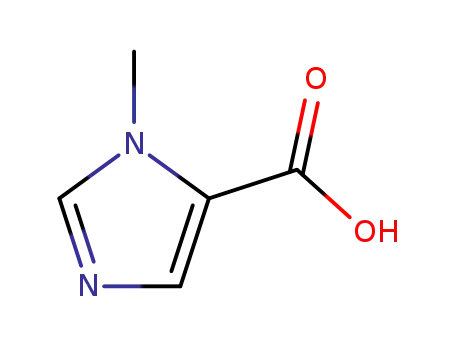
1-methyl-1H-imidazole-5-carboxylic acid
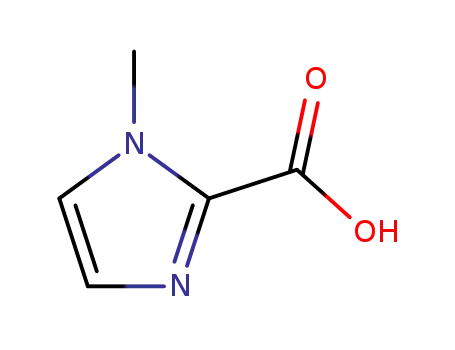
1-methyl-2-imidazolecarboxylic acid
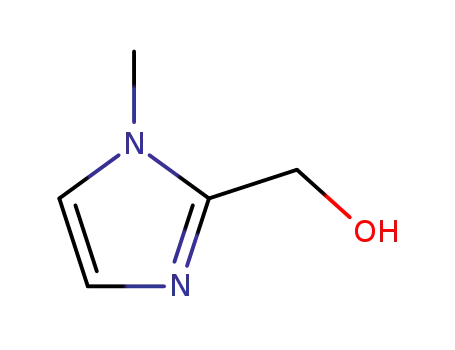
1-methyl-2-hydroxymethylimidazole
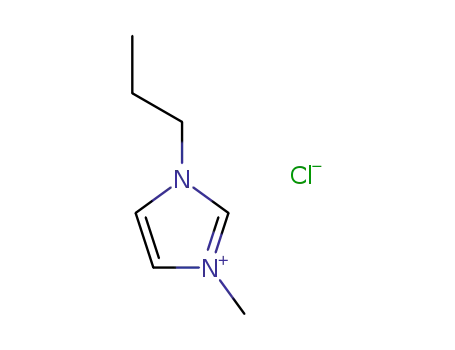
1-methyl-3-propyl-1H-imidazolium chloride