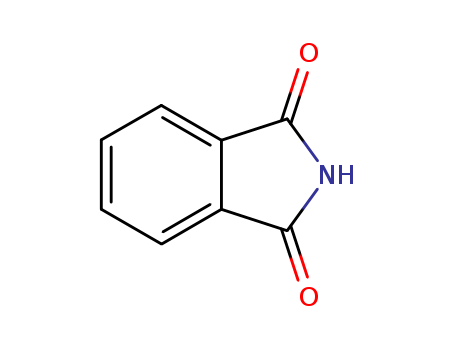


CasNo: 85-41-6
MF: C8H5NO2
Appearance: white to slightly yellowish crystalline flakes
|
Synthesis Reference(s) |
Journal of the American Chemical Society, 111, p. 3725, 1989 DOI: 10.1021/ja00192a034The Journal of Organic Chemistry, 45, p. 363, 1980 DOI: 10.1021/jo01290a038Tetrahedron Letters, 34, p. 6907, 1993 DOI: 10.1016/S0040-4039(00)91827-6 |
|
Air & Water Reactions |
Insoluble in water. |
|
Reactivity Profile |
O-Phthalimide is an imide. Amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx). O-Phthalimide forms salts with bases. |
|
Health Hazard |
ACUTE/CHRONIC HAZARDS: When heated to decomposition O-Phthalimide emits toxic fumes of nitrogen oxides. |
|
Fire Hazard |
Literature sources indicate that O-Phthalimide is combustible. |
|
Safety Profile |
Moderately toxic by intraperitoneal route. Mildly toxic by ingestion. An experimental teratogen. Other experimental reproductive effects. When heated to decomposition it emits toxic fumes of NOx. |
|
Purification Methods |
Crystallise the imide from EtOH (20mL/g) (charcoal), or sublime it. For potassium phthalimide see entry in “Metal-organic Compounds”, Chapter 5. [Beilstein 21/10 V 270.] |
|
Definition |
ChEBI: A dicarboximide that is 2,3-dihydro-1H-isoindole substituted by oxo groups at positions 1 and 3. |
|
General Description |
White to light tan powder. Slightly acidic. |
InChI:InChI=1/C8H5NO2/c10-7-5-3-1-2-4-6(5)8(11)9-7/h1-4H,(H,9,10,11)
The transformation of cyclic hemiacetals...
The ubiquity of nitrogen-containing smal...
A straightforward and general method has...
Abstract: An efficient and simple synthe...
Thalidomide and its derivatives are curr...
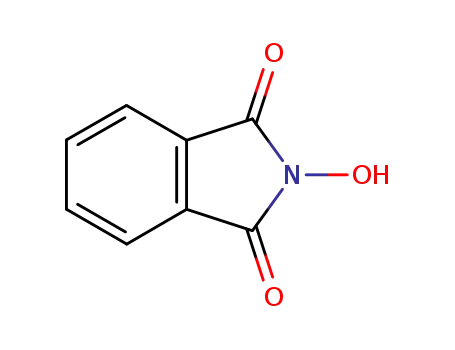
N-hydroxyphthalimide

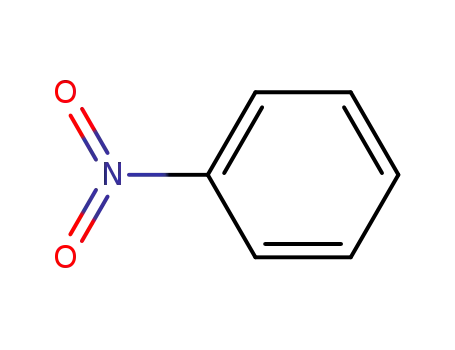
nitrobenzene


phthalimide

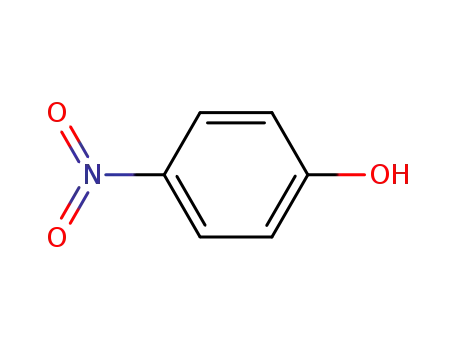
4-nitro-phenol

2-hydroxynitrobenzene
| Conditions | Yield |
|---|---|
|
at 270 ℃; for 30h; Mechanism; also with naphthalene;
|
2-((4-nitrophenoxy)methyl)isoindoline-1,3-dione


phthalimide

formaldehyd


4-nitro-phenol
| Conditions | Yield |
|---|---|
|
With buffer; In methanol; acetonitrile; at 25 ℃; Rate constant; Mechanism; pH 9.0, μ=0.15;
|
N-methylaniline
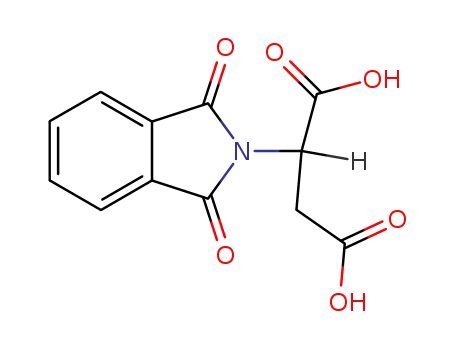
(+/-)-phthaloylaspartic acid
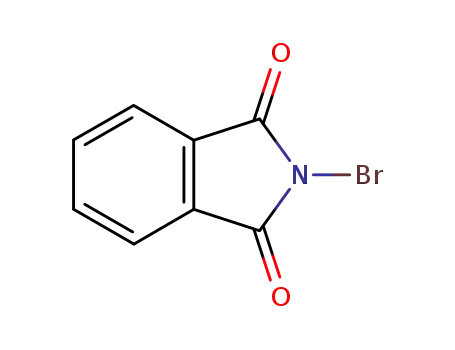
N-bromophthalimide
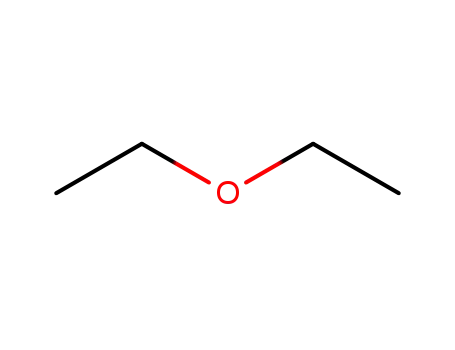
diethyl ether
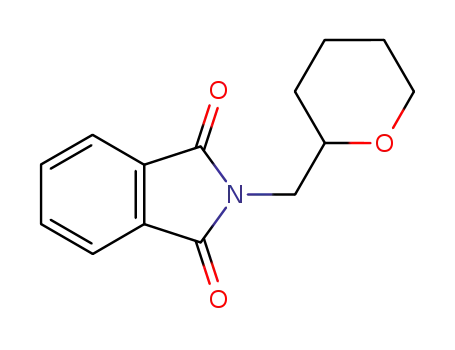
2-(oxan-2-ylmethyl)-2,3-dihydro-1H-isoindole-1,3-dione
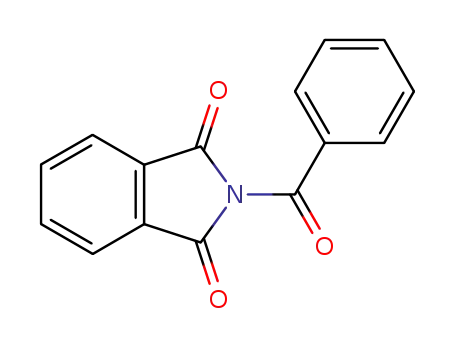
2-benzoylisoindoline-1,3-dione
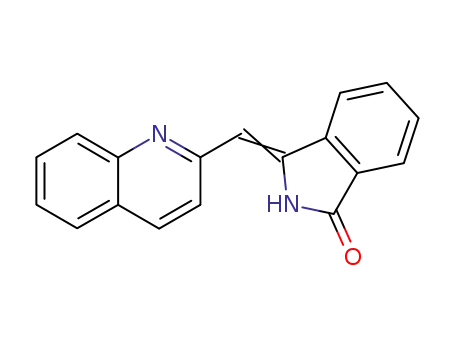
3-quinolin-2-ylmethylene-2,3-dihydro-isoindol-1-one
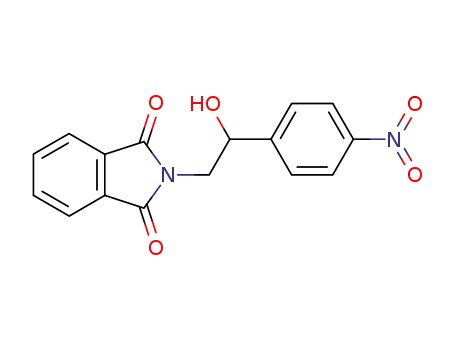
2-(2-hydroxy-2-(4-nitro-phenyl)-ethyl)-isoindole-1,3-dione