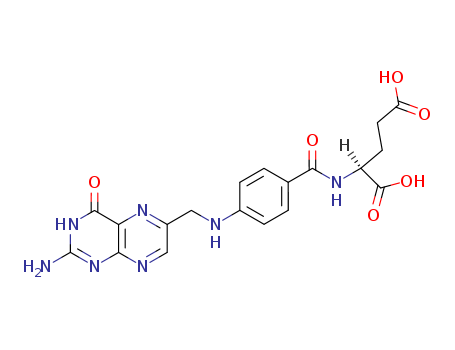


CasNo: 59-30-3
MF: C19H19N7O6
Appearance: orange to yellow crystalline powder
|
Manufacturing Process |
The following description is taken from US Patent 2,956,057. 100 grams of 1,3,3-trichloroacetone are heated on a boiling water bath and 95 grams of bromine are added thereto in drops while being stirred and the stirring is continued for about 1 hour. The resulting reaction solution is distilled under reduced pressure. 115 grams of 1-bromo-1,3,3- trichloroacetone are obtained having a boiling point of 85° to 95°C/17 mm (Hg). For the preparation of the hydrate, 100 grams of water are added to 100 grams of 1bromo-1,3,3-trichloroacetone, which is agitated and cooled. A white scaly crystal of hydrate of 1-bromo-1,3,3-trichloroacetone is obtained (100 grams), having a melting point of 52° to 53°C. 8.9 grams of 2,4,5-triamino-6-hydroxypyrimidine hydrochloride and 8 grams of p-aminobenzoylglutamic acid are dissolved in 400 cc warm water, which is cooled at 35° to 27°C and adjusted to pH 4 by using 20% caustic soda solution. To this solution was simultaneously added dropwise a solution obtained by dissolving 13.4 grams of 1-bromo-1,3,3-trichloroacetone hydrate in 90 cc of 50% methanol and 24 grams of 35% aqueous sodium bisulfite solution over a period of approximately 2 hours. During this period, in order to maintain the pH value of the reaction solution at 4 to 5, 20% caustic soda solution is added from time to time. The precipitate, formed by stirring for 5 hours after dropping was finished, is filtered, and the filtrated precipitate is refined; 5.6 grams of pure pteroylglutamic acid is obtained. |
|
Therapeutic Function |
Treatment of B vitamin (folacin) deficiency |
|
Air & Water Reactions |
Insoluble in water. Aqueous solutions have pHs of 4.0-4.8. |
|
Reactivity Profile |
Acid solutions of Folic acid are sensitive to heat, but towards neutrality, stability progressively increases. Solutions are inactivated by ultraviolet light and alkaline solutions are sensitive to oxidation. Folic acid is also inactivated by light. Folic acid is incompatible with oxidizing agents, reducing agents and heavy metal ions. |
|
Fire Hazard |
Flash point data for Folic acid are not available; however, Folic acid is probably combustible. |
|
Biochem/physiol Actions |
A nutritional delivery form of folate. Folic acid and its derivatives are essential mediators of one-carbon metabolism within cells. |
|
Safety Profile |
Poison by intraperitoneal and intravenous routes. Experimental teratogenic effects. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx. |
|
Veterinary Drugs and Treatments |
Folic acid is used to treat folic acid deficiency in dogs, cats, and horses (theoretically in other animal species as well) often due to small intestinal disease. Cats with exocrine pancreatic insufficiency appear to be most at risk for folate and cobalamin deficiencies secondary to malabsorption of folic acid in the diet. Dogs with exocrine pancreatic insufficiency often are noted to have increased folate levels secondary to overgrowths of folate-synthesizing bacteria in the proximal small intestine. Chronic administration of dihydrofolate reductase inhibiting drugs such as pyrimethamine, ormetoprim or trimethoprim can potentially lead to reduced activated folic acid (tetrahydrofolic acid); folic acid supplementation is sometimes prescribed in an attempt to alleviate this situation. |
|
Drug interactions |
Potentially hazardous interactions with other drugs Antiepileptics: reduces phenytoin, primidone and phenobarbital levels. Cytotoxics: avoid with raltitrexed. |
|
Metabolism |
Folic acid given therapeutically enters the portal circulation largely unchanged, since it is a poor substrate for reduction by dihydrofolate reductase. It is converted to the metabolically active form 5-methyltetrahydrofolate in the plasma and liver. Folate undergoes enterohepatic circulation. Folate metabolites are eliminated in the urine and folate in excess of body requirements is excreted unchanged in the urine. |
|
Purification Methods |
If paper chromatography indicates impurities, then recrystallise it from hot H2O or from dilute acid [Walker et al. J Am Chem Soc 70 19 1948]. Impurities may be removed by repeated extraction with n-BuOH of a neutral aqueous solution of folic acid (by suspending in H2O and adding N NaOH dropwise till the solid dissolves, then adjusting the pH to ~7.0-7.5) followed by precipitation with acid, filtration, or better collected by centrifugation and recrystallised form hot H2O. [Blakley Biochem J 65 331 1975, Kalifa et al. Helv Chim Acta 6 1 2739 1978.] Chromatography on cellulose followed by filtration through charcoal has also been used to obtain pure acid. [Sakami & Knowles Science 129 274 1959.] UV: max 247 and 296nm ( 12,800 and 18,700) in H2O pH 1.0; 282 and 346nm ( 27.600 and 7,200) in H2O pH 7.0; 256, 284 and 366nm ( 24600, 24,500 and 86,00) in H2O pH 13 [Rabinowitz in The Enzymes (Boyer et al. Eds), 2 185 1960]. [Beilstein 26 III/IV 3944.] |
|
Discovery and History |
Folic acid's discovery is attributed to Lucy Wills, a medical researcher who identified its role in preventing severe anemia in pregnant textile workers in India in 1930. Initially known as "the Wills Factor," it was later designated as vitamin M, vitamin Bc, Lactobacillus casei growth factor, and eventually vitamin B9. Folic acid was isolated from spinach in 1941, and its industrial synthesis was developed in 1945. |
|
Chemical Structure |
Folic acid has the chemical formula C19H19N7O6 and consists of a heterocyclic pterin structure with a methyl group in the sixth position bound to para-aminobenzoic and glutamic acids. It is also known as pteroylglutamic acid and belongs to the folate group, which includes its derivatives like dihydro-, tetrahydro-, methyl-, and formyl-compounds conjugated with para-aminobenzoyl-glutamate. |
|
Biological Functions |
Folic acid plays a crucial role in various biological processes, including early neurodevelopment and the prevention of neural tube defects, particularly spina bifida. It is involved in homocysteine metabolism, where supplementation can lower serum homocysteine levels and reduce the risk of ischemic heart disease and stroke. National health agencies recommend daily supplementation of folic acid for women of childbearing age to reduce the risk of neural tube defects during pregnancy. |
|
Fortification and Recommendations |
The US Public Health Service recommends that all individuals capable of becoming pregnant consume 400 渭g/day of folic acid to prevent neural tube defects. Fortification of foods with folic acid has been recognized as an effective strategy in populations with limited access to folic acid-rich foods or supplements, ensuring adequate intake for neural tube defect prevention. |
|
Physical properties |
The folates include a large number of chemically related species, each differing with respect to the various substituents possible at three sites on the pteroylglutamic acid basic structure. More than 170 different folates are theoretically possible. Not all of these occur in nature; but it has been estimated that as many as 100 different forms are found in animals. The folates from most natural sources usually have a single carbon unit at N-5 and/or N-10; these forms participate in the metabolism of the single-carbon pool.Folic acid (pteroylmonoglutamic acid) is an orange-yellow crystalline substance that is soluble in water but insoluble in ethanol or less polar organic solvents. It is unstable to light, to acidic or alkaline conditions, to reducing agents, and, except in dry form, to heat. It is reduced in vivo enzymatically (or in vitro with a reductant such as dithionite) first to 7,8-dihydrofolic acid (FH2) and then to FH4; both of these compounds are unstable in aerobic environments and must be protected by the pres ence of an antioxidant (e.g., ascorbic acid, 2-merTwo derivatives of folic acid, each having an amino group in the place of the hydroxyl at C-4, are folate antagonists of biomedical use: aminopterin (4-aminofolic acid) and methotrexate (4-amino-N10-methylfolic acid). Aminopterin is used as a rodenticide; methotrexate is an antineoplastic agent. |
|
Definition |
ChEBI: An N-acyl-amino acid that is a form of the water-soluble vitamin B9. Its biologically active forms (tetrahydrofolate and others) are essential for nucleotide biosynthesis and homocysteine remethylation. |
|
General Description |
Odorless orange-yellow needles or platelets. Darkens and chars from approximately 482°F. |
InChI:InChI=1/C19H19N7O6/c20-19-25-15-14(17(30)26-19)23-11(8-22-15)7-21-10-3-1-9(2-4-10)16(29)24-12(18(31)32)5-6-13(27)28/h1-4,8,12,21H,5-7H2,(H,24,29)(H,27,28)(H,31,32)(H3,20,22,25,26,30)/p-2/t12-/m1/s1
-
Abstract: Composition of biligand compou...
The invention belongs to the technical f...
The invention relates to a preparation m...
The invention discloses a preparation me...
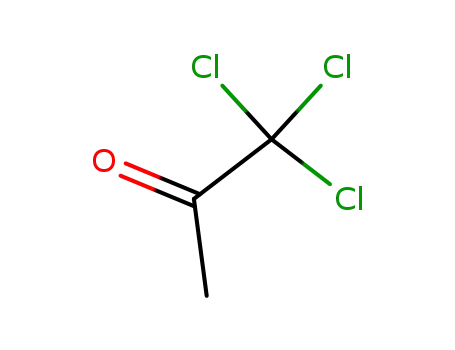
1,1,1-trichloroacetone

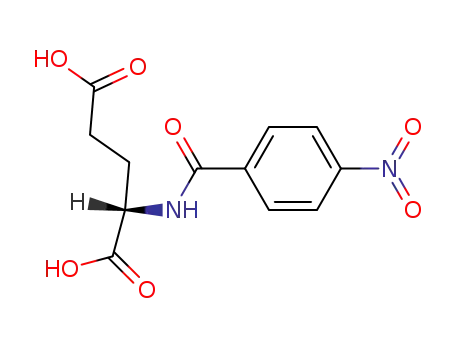
p-Nitrobenzoyl-L-(+)-glutamic acid

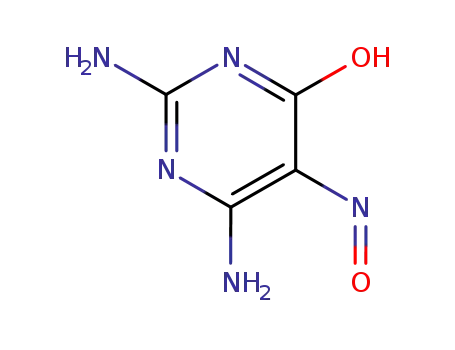
2,4-diamino6-hydroxy-5-nitroso pyrimidine

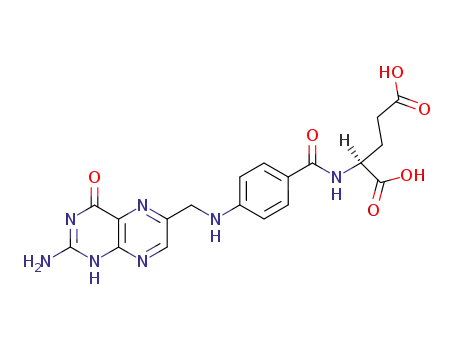
folate
| Conditions | Yield |
|---|---|
|
p-Nitrobenzoyl-L-(+)-glutamic acid; 2,4-diamino6-hydroxy-5-nitroso pyrimidine; With hydrogen; In water; at 82 ℃; under 6000.6 Torr;
1,1,1-trichloroacetone; With sodium metabisulfite; sodium hydroxide; In water; at 45 ℃; pH=4.5; pH-value; Pressure; Temperature;
|
80% |

1,1,1-trichloroacetone

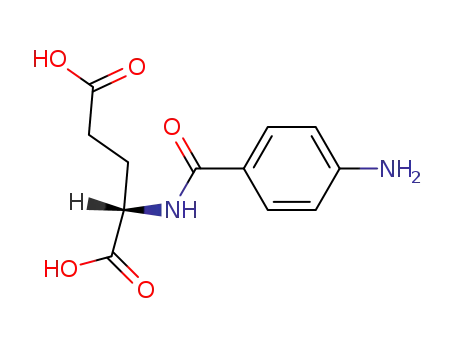
N-(4-aminobenzoyl)-L-glutamic acid

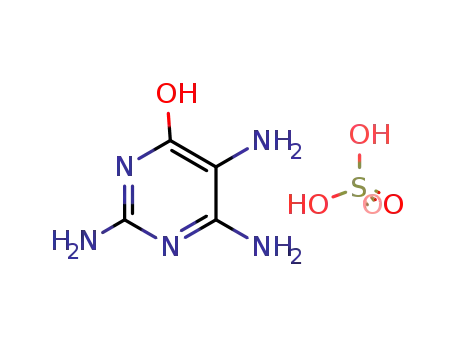
2,4,5-triamino-6-hydroxypyrimidine sulfate


folate
| Conditions | Yield |
|---|---|
|
1,1,1-trichloroacetone; N-(4-aminobenzoyl)-L-glutamic acid; With sodium acetate; sodium sulfite; In acetic acid; at 43 ℃; for 0.5h;
2,4,5-triamino-6-hydroxypyrimidine sulfate; In acetic acid; at 38 ℃; for 5h;
|
75.5% |
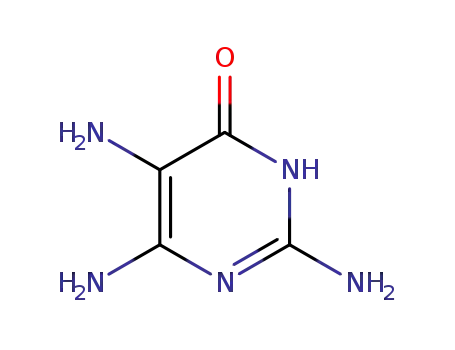
2,5,6-triamino-3,4-dihydro-4-pyrimidinone
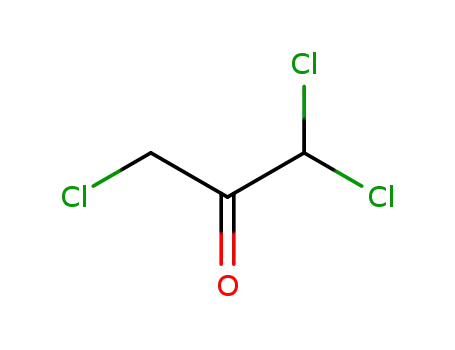
1,1,3-trichloroacetone

N-(4-aminobenzoyl)-L-glutamic acid
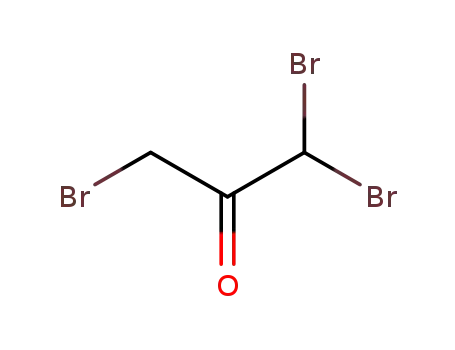
1,1,3-tribromoacetone
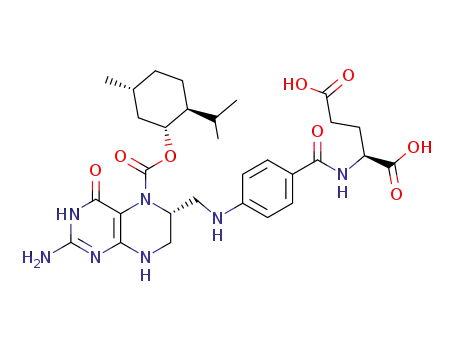
5-(-)-menthyloxycarbonyl-(6R)-tetrahydrofolic acid
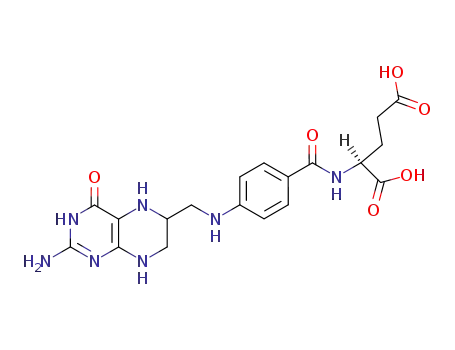
tetrahydrofolic acid
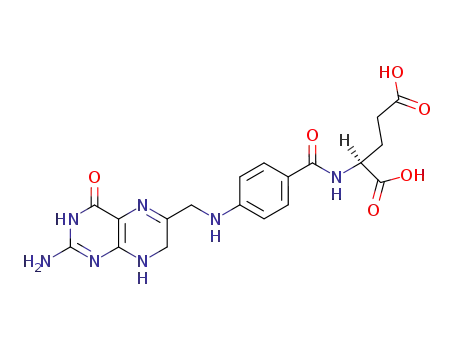
(S)-2-{4-[(2-Amino-4-oxo-3,4,7,8-tetrahydro-pteridin-6-ylmethyl)-amino]-benzoylamino}-pentanedioic acid
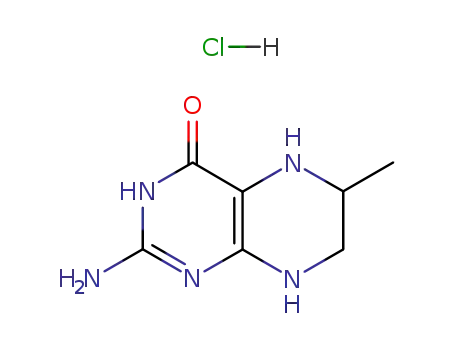
6-methyl-5,6,7,8-tetrahydropterin hydrochloride