

CasNo: 66-22-8
MF: C4H4N2O2
Appearance: white powder
|
Organic alkali |
Uracil is an organic alkali, and is one of the four major bases in RNA. It is a major component of the pyrimidine composition in ribonucleic acid (RNA) as well as in various kinds of uridines. It can connect with ribose to generate UMP whose triphosphate compound being UTP. UTP is the precursor form of uracil in RNA biosynthesis. UTP also acts as a coenzyme to be involved in the biosynthesis of certain sugars. Uracil can block the degradation effect of tegafur, and thus increasing the concentration of fluorouracil which enhance the anti-cancer effects. Fluorouracil has similar clinical indications as uracil. It is mainly used for treating digestive cancer, breast cancer and thyroid cancer. Combination with mitomycin has a good efficacy on treating advanced gastric cancer. Laboratory synthesizes uracil through the cyclization reaction between ethyl malonyl and urea for pharmaceutical and biochemical research. Uracil has tautomerism effect: Keto (2,4-2 CPCC) enol (2,4-2-hydroxy pyrimidine) in mainly exist in the form of ketone inside biological cells. Nature uracil is presented mainly in marine organisms, particulate matter and sea lysate. It is treated as life indicator in the field of organic geochemistry. Pyrimidine refers to the hexaheterocyclic compound with two nitrogen atoms in 1,3-position of the benzene ring, and it, together with pyridazine and pyrazine, are isomers of each other. Pyrimidine has a unique UV spectrum due to the presence of conjugated double bonds in its structure. Pyrimidine has a lower basicity and a weaker lectrophilic substitution reaction than pyridine. But it is more prone to have nucleophilic substitution. Derivatives of pyrimidine are widely distributed in nature, including vitamin B1, uracil, thymine, and cytosine which all containing a pyrimidine structure. |
|
Fluorouracil |
Fluorouracil, briefly referred as FU, is currently one of the most commonly used anti-cancer drug. It is white crystals with pKa = 8.1, m.p.282~283 °C. It is slightly soluble in water (12mg/ml at 25 °C) and ethanol, but insoluble in chloroform and ether. It is easily soluble in diluted acid and alkali. It is hydrolyzed in the presence of strong base but is stable in normal saline. Due to the introduction of a strong electrically fluorine atoms, the acidity of Fu is 30 times higher than its parent, uracil. The injection of Fu usually is an aqueous solution with pH 9.0 adjusted by sodium hydroxide. It is sensitive to light and easy to crystallize when stored at low temperatures or prolonged room temperature. According to the stronger ability of tumor tissue of rats in utilizing pyrimidine than normal tissue n, in 1957, Duschinsky and Heidelbergere designed and replace the 5-hydrogen in uracil to fluorine with similar size and generated Fu, as an anti-metabolite of uracil to achieve selective anticancer effects. FU has inhibitory effects on many kinds of animal transplanted tumors such as mouse leukemia L1210, L615, and adenocarcinoma 755. Tumor cells has no cross-resistance to it and other commonly used anti-cancer drugs such as cytarabine, methotrexate, mercaptopurine, cyclophosphamide, and carmustine. FU is converted into 5-fluoro-deoxy-uridine monophosphate (FDUMP) and 5-fluorouracil nucleoside triphosphate (FUTP) in tissues. FDUMP inhibits the thymidylate synthase (TS) via forming compound with TS and 5,10-methenyltetrahydrofolate, thus resulting in a lack of intracellular thymine nucleotide and further inhibition of DNA synthesis, finally leading to cell death. On the other hand, FUTP is incorporated into RNA as the substrate of RNA polymerase substrate and affect the normal synthesis and function of RNA. In tissue culture, supplement of thymidine (TdR) can reverse the FU cytotoxicity, so that it has been realized for many years that the impact on DNA is the primary growth-inhibitory mechanism of FU. However, it was found that TdR didn’t completely reverse the cytotoxicity of FU, and the combination of FU and TdR significantly improved the FU’s incorporation into RNA and its anti-cancer effect. After culturing together of L1210 leukemia cells with 6-H3-5FU for 22 hours, it was found the existence of 80 fmol of FDUMP-TS-5,10CH2-H4 folic acid complexes in 106 cell while 400 fmol of FU bound to RNA. This emphasizes the importance of FU’s incorporation into RNA FU for its anti-tumor effect. FU is a cell cycle-specific drug which playing the significant role at S-phase. Reference: China Medical Encyclopedia Editing Committee; editor: Liang Huang; Chinese Medical Encyclopedia. The above information is edited by the lookchem of Dai Xiongfeng. |
|
Chemical property |
White or light yellow crystalline needle. Melting point 338 °C; easily soluble in water, soluble in diluted ammonia, slightly soluble in cool water, insoluble in alcohol and ether. |
|
Production methods |
It is produced through the reaction of malate, sulfuric acid and urea. |
|
Safety Profile |
Moderately toxic by intraperitoneal route. An experimental teratogen. Experimental reproductive effects. Questionable carcinogen with experimental tumorigenic data. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx. |
|
Purification Methods |
Uracil crystallises from water (m 339-341o) and m 338o after sublimation in high vacuum. Its solubility in H2O at 20o is 1g/300mL. [Beilstein 24 H 312, 24 I 312, 24 II 169, 24 III/IV 1193.] |
|
Used in Medicinal Chemistry |
Uracil and its derivatives are integral components of numerous commercial drugs with therapeutic potential in countering various pathogenic and physiological disorders. These compounds are employed in the treatment of viral infections, cancer, diabetes, thyroid disorders, and autosomal recessive disorders. Uracil-based drugs inhibit RNA viruses like influenza virus and coxsackievirus B4, and they demonstrate antibacterial, antifungal, antimicrobial, antitubercular, and antiprotozoal properties. Additionally, uracil derivatives have shown efficacy against pathogens such as Trichomonas vaginalis, Trypanosoma brucei, Leishmania mexicana, and Trypanosoma cruzi. The pharmacological activities of uracil compounds are attributed to their interactions with specific molecular targets involved in disease pathways, making them valuable candidates for drug discovery and development. [1] |
|
Used in Perovskite Solar Cells |
Uracil is utilized in the fabrication of high-performance perovskite solar cells to improve both power conversion efficiency (PCE) and operational stability. When introduced as a "binder" into the perovskite film, uracil efficiently passivates defects and strengthens grain boundaries, enhancing the stability of perovskite films. Additionally, uracil strengthens the interface between the perovskite and the Tin oxide (SnO2) electron transport layer, increasing the binding force. These modifications result in perovskite solar cells with superior operational stability, delivering high PCE and maintaining over 90% of their initial PCE even after extended exposure to continuous light. [1] |
|
Production Methods |
Uracil can be synthesized through various methods, including the deamination of cytosine and synthetic routes involving urea, thiourea, maleic acid, and fumaric acid. [1] |
|
Definition |
A nitrogenous base that is found in RNA, replacing the thymine of DNA. It has a pyrimidine ring structure. |
|
General Description |
Certified pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards. Uracil is one of the nucleotide bases of RNA. It is the precursor of DNA′s thymine. It acts like a carrier of genetic data and is hooked up with a ribose and three phosphates to form a ribonucleoside triphosphate once a human body produces nucleotides. |
InChI:InChI=1/C4H4N2O2/c7-3-1-2-5-4(8)6-3/h1-2H,(H2,5,6,7,8)
We report two-dimensional (2D) 17O multi...
The invention relates to a green prepara...
The invention relates to the field of ch...
The poor solubility of many nucleosides ...
We recently discovered a [Fe-S]-containi...
2,6 dichloropurine

uridine

uracil

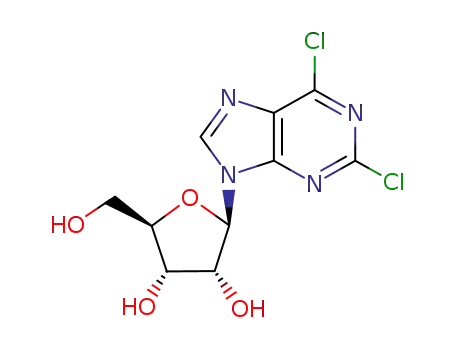
2,6-dichloropurine riboside
| Conditions | Yield |
|---|---|
|
With potassium phosphate; at 40 ℃; for 6h; pH=7.5; Equilibrium constant; Enzymatic reaction;
|
34 mg |
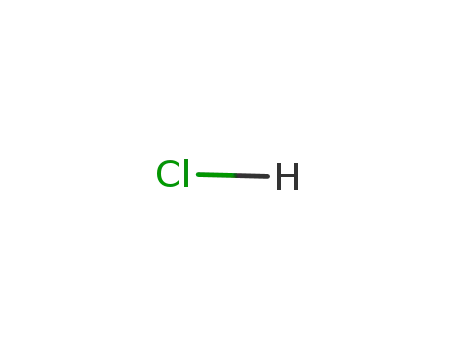
hydrogenchloride

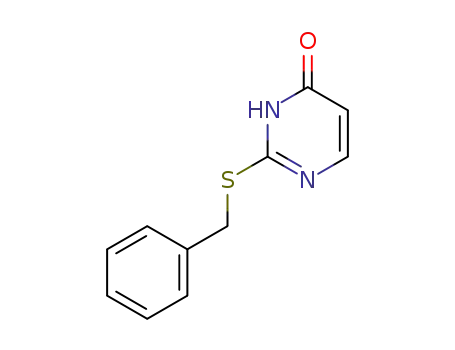
2-(benzylsulfanyl)pyrimidin-4(3H)-one


phenylmethanethiol


uracil
| Conditions | Yield |
|---|---|
|
|

uracil
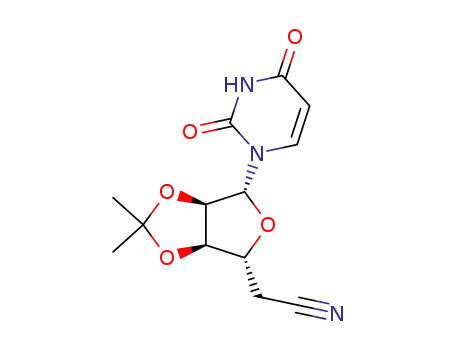
5'-Desoxy-2',3'-O-isopropylidenuridin-5'-carbonitril
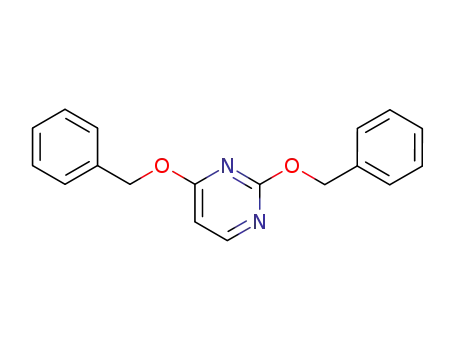
2,4-bis(benzyloxy)pyrimidine
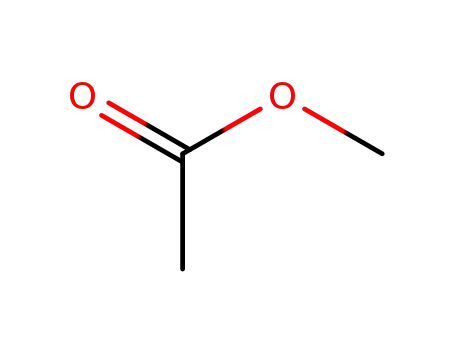
acetic acid methyl ester
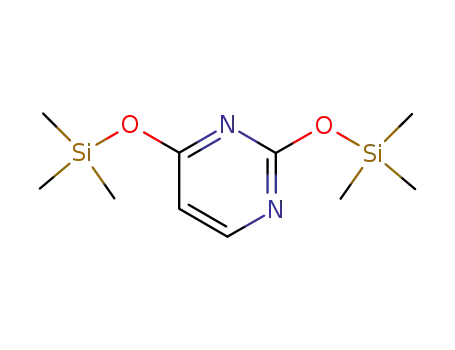
O,O'-bis-(trimethylsilyl)uracil
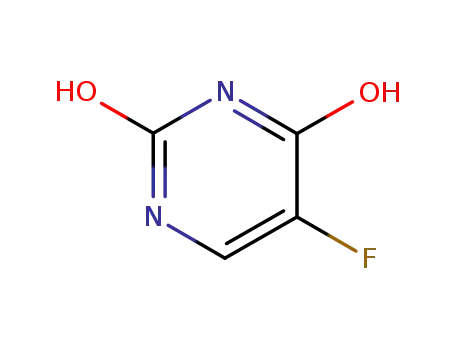
5-fluoropyrimidine-2,4-diol
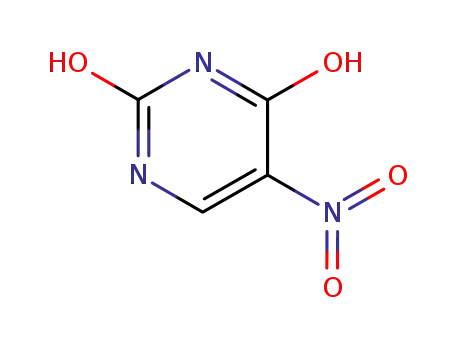
2,4-dihydroxy-5-nitropyrimidine
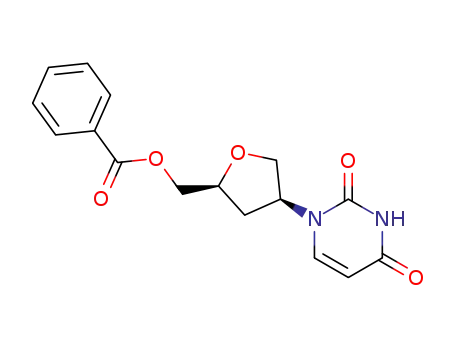
4(S)-<3,4-dihydro-2,4-dioxo-1(2H)-pyrimidinyl>tetrahydro-2(S)-furanmethanol benzoate