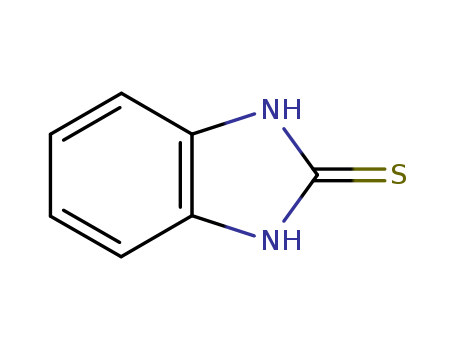


CasNo: 583-39-1
MF: C7H6N2S
Appearance: White to yellow crystals or cream colored powder
|
Chemical Description |
2-mercaptobenzimidazole is a heterocyclic compound containing a benzimidazole ring and a thiol group. |
|
Reference |
Manohar, D. M., K. A. Krishnan, and T. S. Anirudhan. "Removal of mercury (II) from aqueous solutions and chlor-alkali industry wastewater using 2-mercaptobenzimidazole-clay." Water Research 36.6(2002):1609. Chadwick, D., and T. Hashemi. "Electron spectroscopy of corrosion inhibitors: Surface films formed by 2-mercaptobenzothiazole and 2-mercaptobenzimidazole on copper." Surface Science 89.1(1979):649-659. Trachli, B., et al. "Protective effect of electropolymerized 2-mercaptobenzimidazole upon copper corrosion." Progress in Organic Coatings 44.1(2002):17-23. Wang, Lin. "Evaluation of 2-mercaptobenzimidazole as corrosion inhibitor for mild steel in phosphoric acid." Corrosion Science 43.12(2001):2281-2289. Mazloum, M, M. K. Amini, and I. Mohammadpoor-Baltork. "Mercury selective membrane electrodes using 2-mercaptobenzimidazole, 2-mercaptobenzothiazole, and hexathiacyclooctadecane carriers." Sensors & Actuators B Chemical 63.1(2000):80-85. https://www.alfa.com/en/catalog/A18350/ |
|
Synthesis Reference(s) |
Organic Syntheses, Coll. Vol. 4, p. 569, 1963The Journal of Organic Chemistry, 29, p. 3209, 1964 DOI: 10.1021/jo01034a020 |
|
Air & Water Reactions |
Dust explosion: 0.140 oz/ft3. Insoluble in water. 2-Mercaptobenzimidazole is moisture sensitive. . |
|
Reactivity Profile |
An organosulfide and an amine. Organosulfides are incompatible with acids, diazo and azo compounds, halocarbons, isocyanates, aldehydes, alkali metals, nitrides, hydrides, and other strong reducing agents. Reactions with these materials generate heat and in many cases hydrogen gas. Many of these compounds may liberate hydrogen sulfide upon decomposition or reaction with an acid. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. |
|
Fire Hazard |
Flash point data for 2-Mercaptobenzimidazole are not available; however, 2-Mercaptobenzimidazole is probably combustible. |
|
Flammability and Explosibility |
Nonflammable |
|
Safety Profile |
Poison by intraperitoneal and intravenous routes. Moderately toxic by ingestion. Skin and eye irritant. When heated to decomposition it emits toxic fumes of SO, and NO,. See also MERCAPTANS. |
|
Purification Methods |
Crystallise it from aqueous EtOH, AcOH or aqueous ammonia. It complexes with many metals. [Brown J Chem Soc 1976 1958, Beilstein 24 II 65, 24 III/IV 287.] |
|
General Description |
White to yellow crystals or cream colored powder. Slight odor. |
InChI:InChI=1/C7H6N2S/c10-9-5-8-6-3-1-2-4-7(6)9/h1-5,10H
A green and efficient method has been de...
A practical and efficient method to synt...
-
A concise and versatile method for the c...
lansoprasole

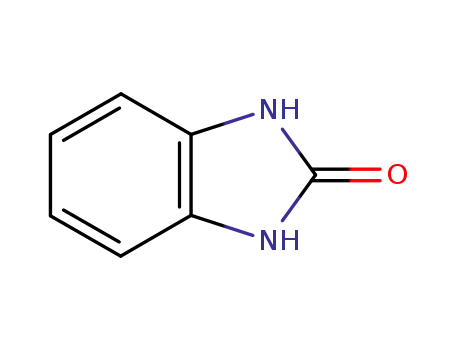
1,3-dihydro-2H-benzimidazol-2-one

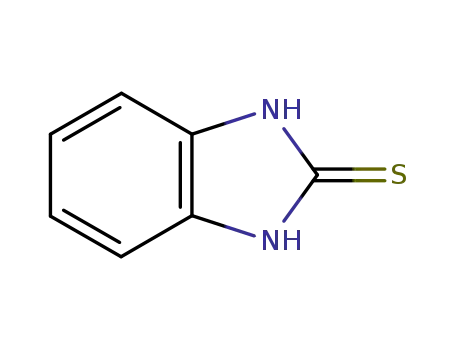
2,3-dihydrobenzimidazol-2-thione
| Conditions | Yield |
|---|---|
|
With water; at 40 ℃; for 5844h;
|
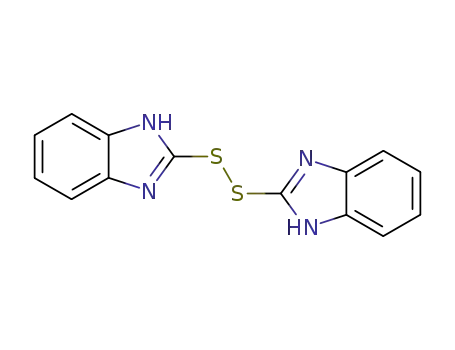
bis-benzimidazole disulfide

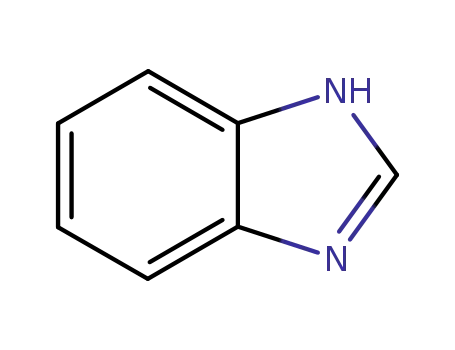
benzoimidazole


1,3-dihydro-2H-benzimidazol-2-one


2,3-dihydrobenzimidazol-2-thione
| Conditions | Yield |
|---|---|
|
With oxygen; In acetonitrile; for 120h; Mechanism; Product distribution; Irradiation;
|
11.2% 8.6% 21% |
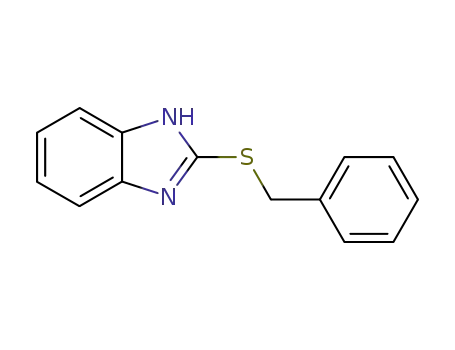
2-(benzylthio)-1H-benzo[d]imidazole
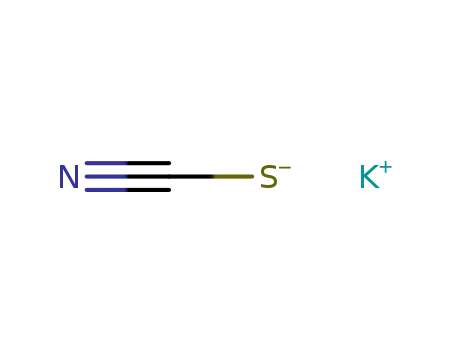
potassium thioacyanate
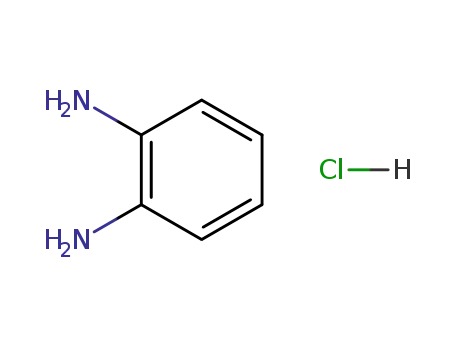
o-phenylenediamine hydrochloride
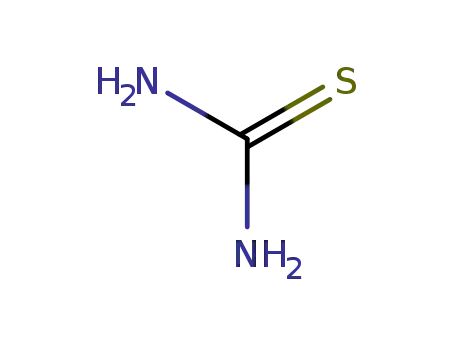
thiourea
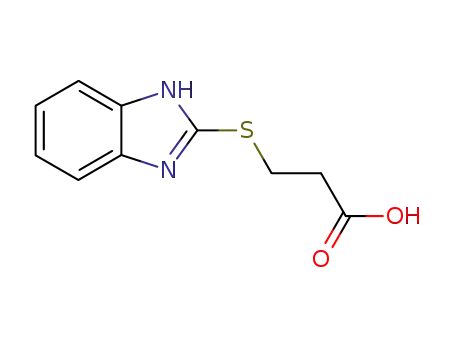
Benzimidazolyl-2-thiopropionic Acid
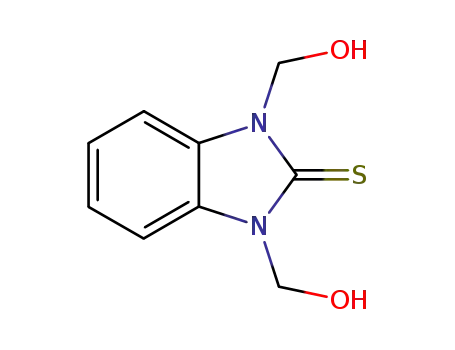
1,3-di(hydroxymethyl)-2,3-dihydro-1H-benzo[d]imidazole-2-thione
2-(2-benzimidazolylthio)propionic acid

2-(benzylthio)-1H-benzo[d]imidazole