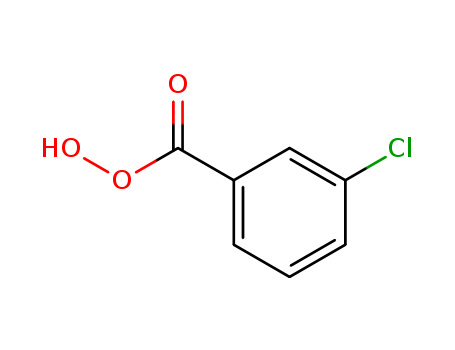


CasNo: 937-14-4
MF: C7H5ClO3
Appearance: white powder
|
Reactions |
3-Chloroperoxybenzoic acid (MCPBA) is one of the most popular oxidation reagent in organic synthesis, because of its outstanding performance in terms of: reactivity, combined with reducing the number of reaction steps in classical synthetic routes, regio- and stereoselectivity, protection of functional groups mostly not required, high purity and yields. Its literature covers a huge area of different syntheses and below reaction equations just can be a brief overview of its interesting applications: |
|
Synthesis Reference(s) |
Synthetic Communications, 19, p. 1271, 1989 DOI: 10.1080/00397918908054534 |
|
General Description |
3-Chloroperoxybenzoic acid is sensitive to heat. Storage of 3-Chloroperoxybenzoic acid must be done so with stringent temperature control measures. It's explosion hazard is also mitigated by mixing the peroxide in a solvent slurry. |
|
Reactivity Profile |
May explode from heat, shock, friction or contamination. May ignite combustibles (wood, paper, oil, clothing, etc.). May be ignited by heat, sparks or flames. |
|
Purification Methods |
Recrystallise MCPBA from CH2Cl2 [Traylor & Mikztal J Am Chem Soc 109 2770 1987]. Peracid of 99+% purity can be obtained by washing commercial 85% material with phosphate buffer pH 7.5 and drying the residue under reduced pressure. Alternatively the peracid can be freed from m-chlorobenzoic acid by dissolving 50g/L of *benzene and washing with an aqueous solution buffered at pH 7.4 (NaH2PO4/NaOH) (5 x 100mL). The organic layer is dried over MgSO4 and carefully evaporated under vacuum. Necessary care should be taken in case of EXPLOSION. The solid is recrystallised twice from CH2Cl2/Et2O and stored at 0o in a plastic container as glass catalyses the decomposition of the peracid. The acid is assayed iodometrically. [Schwartz & Blumbrgs J Org Chem 29 1976 1964, Bortolini et al. J Org Chem 52 5093 1987, McDonald et al. Org Synth Coll Vol VI 276 1988, Beilstein 9 IV 972.] |
InChI:InChI:1S/C7H5ClO3/c8-6-3-1-2-5(4-6)7(9)11-10/h1-4,10H
The invention discloses a method for pre...
The method comprises the following steps...
The invention discloses a synthetic meth...
Abstract: A heterogeneous bifunctional h...

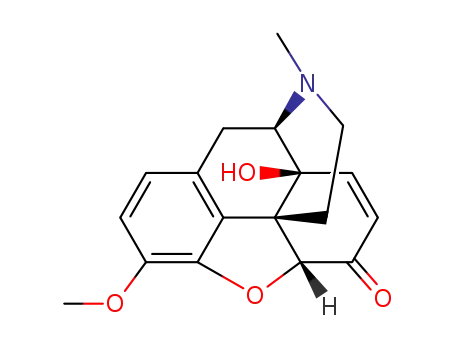
14-hydroxycodeinone


3-chloro-benzenecarboperoxoic acid
| Conditions | Yield |
|---|---|
|
|
72% |
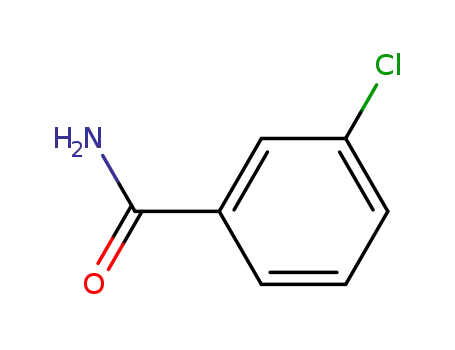
3-chlorobenzamide


3-chloro-benzenecarboperoxoic acid
| Conditions | Yield |
|---|---|
|
3-chlorobenzamide; With sodium nitrite; N-bromoacetamide; at 16 ℃; for 2.16667h;
With zinc fluoride; water; for 4h; Temperature;
|
98.7% |
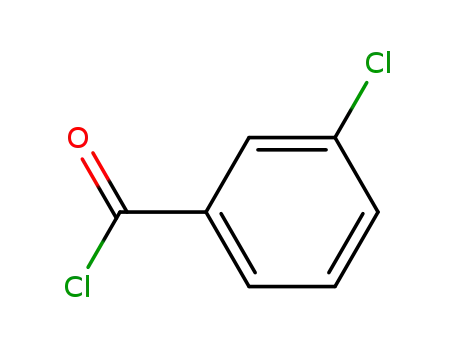
m-Chlorobenzoyl chloride
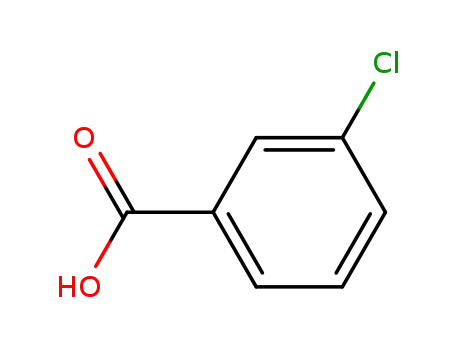
3-chlorobenzoate
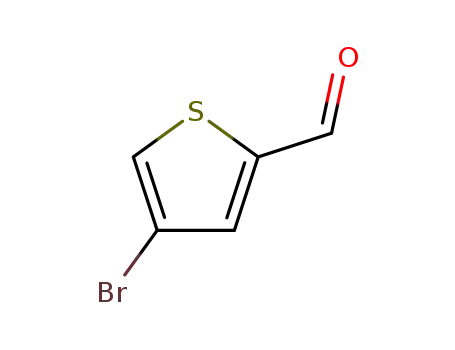
4-Bromothiophen-2-aldehyde
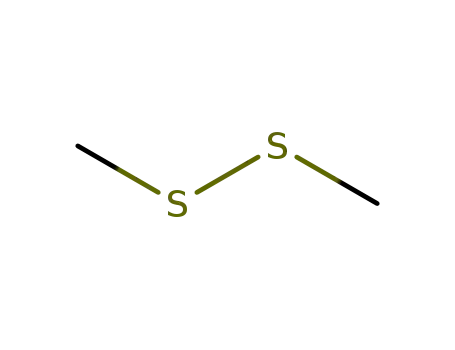
Dimethyldisulphide

N,N-dimethyl-4-(phenyl-trans-azo)-aniline-N-oxide
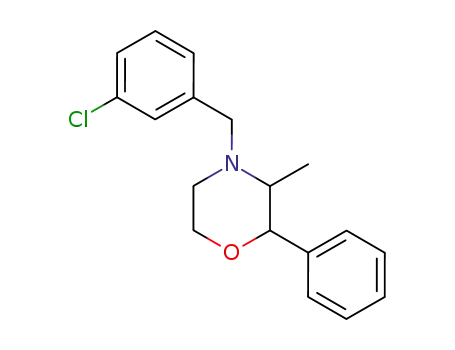
4-(3-chloro-benzyl)-3-methyl-2-phenyl-morpholine
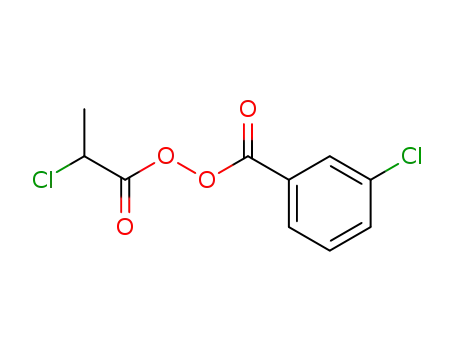
α-Chlorpropionyl-m-chlorbenzoylperoxid
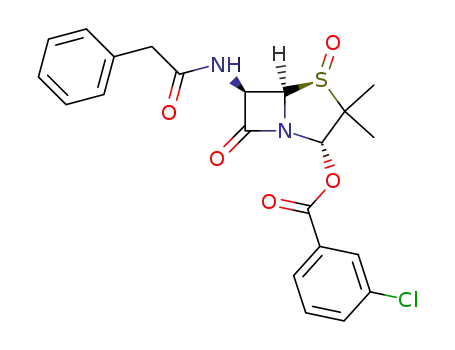
N-[(5R)-2c-(3-chloro-benzoyloxy)-3,3-dimethyl-4t,7-dioxo-(5rH)-4λ4-thia-1-aza-bicyclo[3.2.0]hept-6t-yl]-2-phenyl-acetamide