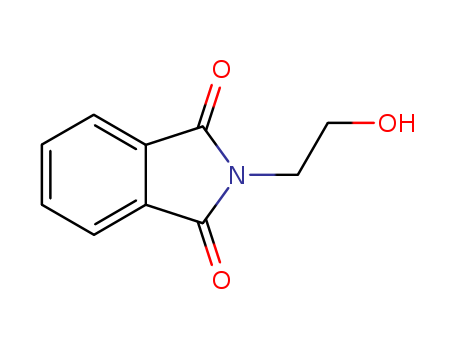
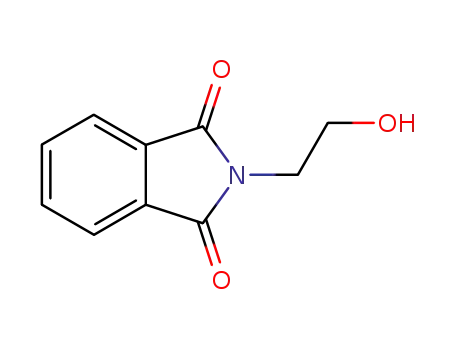
Trustworthy Factory Supply 3891-07-4 with Lowest Price, Factory Sells N-Hydroxyethylphthalimide
- Molecular Formula:C10H9NO3
- Molecular Weight:191.186
- Appearance/Colour:white crystalline powder
- Vapor Pressure:1.06E-05mmHg at 25°C
- Melting Point:126-129 °C
- Refractive Index:1.622
- Boiling Point:356.5 °C at 760 mmHg
- PKA:14.40±0.10(Predicted)
- Flash Point:169.4 °C
- PSA:57.61000
- Density:1.388 g/cm3
- LogP:0.21280
N-Hydroxyethylphthalimide(Cas 3891-07-4) Usage
|
Synthesis Reference(s)
|
Organic Syntheses, Coll. Vol. 4, p. 106, 1963The Journal of Organic Chemistry, 24, p. 1122, 1959 DOI: 10.1021/jo01090a601
|
|
General Description
|
N-(2-Hydroxyethyl)phthalimide is the precursor for chloromethyl ethers used in the synthesis of purine acyclic nucleosides.
|
InChI:InChI=1/C10H9NO3/c12-6-5-11-9(13)7-3-1-2-4-8(7)10(11)14/h1-4,12H,5-6H2
3891-07-4 Relevant articles
Microgels as Matrices for Molecular Receptor and Reactive Sites: Synthesis and Reactivity of Cavities possessing Amino-functions
Hopkins, Andrew,Williams, Andrew
, p. 891 - 896 (1983)
Non-fluxional polymeric particles, micro...
Activities of 2-phthalimidethanol and 2-phthalimidethyl nitrate, phthalimide analogs devoid of the glutarimide moiety, in experimental models of inflammatory pain and edema
Godin, Adriana M.,Araújo, Débora P.,Menezes, Raquel R.,Brito, Ana Mercy S.,Melo, Ivo S.F.,Coura, Giovanna M.E.,Soares, Darly G.,Bastos, Leandro F.S.,Amaral, Flávio A.,Ribeiro, Lucas S.,Boff, Daiane,Santos, Julliana R.A.,Santos, Daniel A.,Teixeira, Mauro M.,De Fátima, ?ngelo,Machado, Renes R.,Coelho, Márcio M.
, p. 291 - 298 (2014)
The reintroduction of thalidomide in the...
-
Wenker
, p. 422 (1937)
-
Facile synthesis of indolizinoindolone, indolylepoxypyrrolooxazole, indolylpyrrolooxazolone and isoindolopyrazinoindolone heterocycles from indole and imide derivatives
Argade, Narshinha P.,Shelar, Santosh V.
, p. 6160 - 6169 (2021/07/21)
Chemo-, regio- and diastereoselective co...
Novel synthesis method of N-hydroxyethyl phthalimide
-
Paragraph 0014-0023, (2021/09/04)
The invention discloses a novel synthesi...
FLUORINATED BENZO[F]BENZIMIDAZOL-4-9-DIONE IUM DERIVATIVES AND PHARMACEUTICAL COMPOSITIONS THEREOF AND THEIR USE AS SURVIVIN SUPPRESSANTS
-
Paragraph 00176; 00178, (2020/09/30)
The present application relates to novel...
Synthesis method of 2-(phthalimido)ethanesulfonyl chloride
-
Paragraph 0033; 0034; 0037; 0038; 0041; 0042; 0045; 0046, (2019/03/28)
The invention discloses a synthesis meth...
3891-07-4 Process route
-
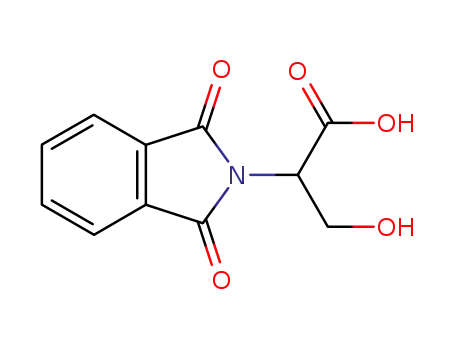
- 65391-10-8
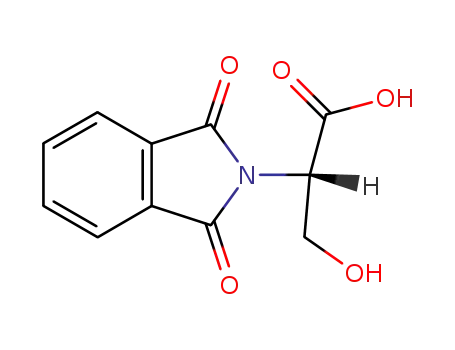
N-phthalimido-D,L-serine
-
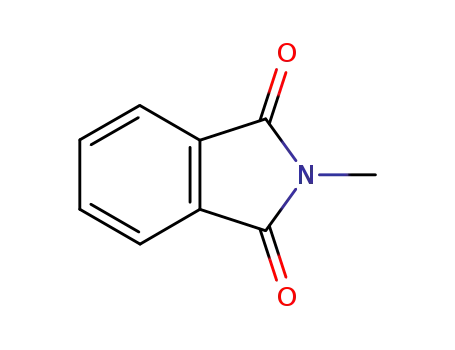
- 550-44-7
N-methylphthalimide
-
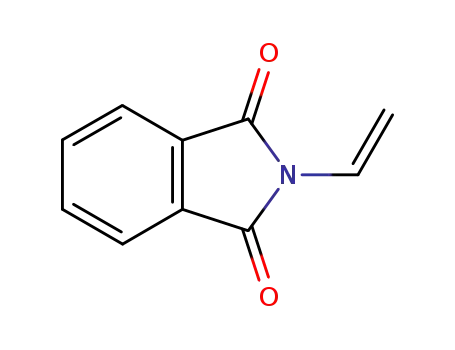
- 3485-84-5,26809-43-8
N-vinylphthalimide
-
- 3891-07-4
N-(2-Hydroxyethyl)phthalimide
Conditions
| Conditions |
Yield |
|
In acetonitrile; for 10h; Irradiation;
|
75%
10% |
-
- 29588-89-4
(2S)-2-(1,3-dioxoisoindolin-2-yl)-3-hydroxypropanoic acid
-
- 550-44-7
N-methylphthalimide
-
- 3485-84-5,26809-43-8
N-vinylphthalimide
-
- 3891-07-4
N-(2-Hydroxyethyl)phthalimide
Conditions
| Conditions |
Yield |
|
In acetonitrile; at 13 ℃; for 24h; Product distribution; Mechanism; Irradiation; effect of various solvents on product/yield distribution;
|
83 % Spectr.
9 % Spectr.
8 % Spectr. |
3891-07-4 Upstream products
-
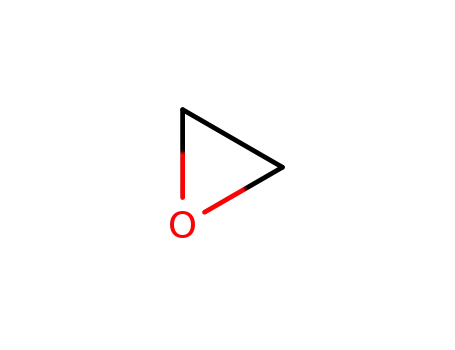
75-21-8
oxirane
-
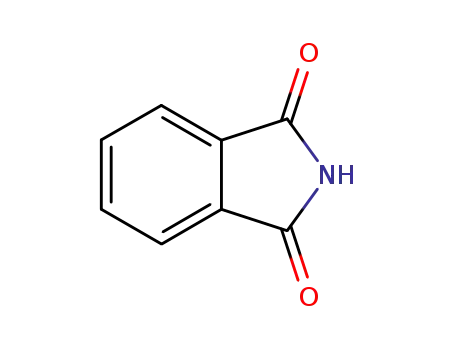
136918-14-4
phthalimide
-
96-49-1
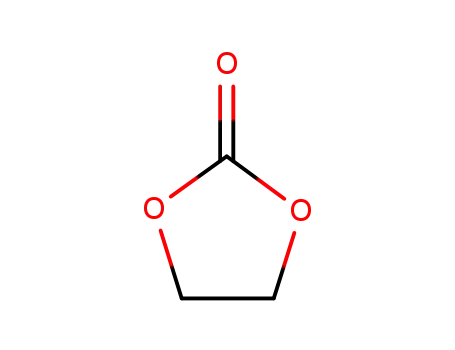
[1,3]-dioxolan-2-one
-
85-44-9
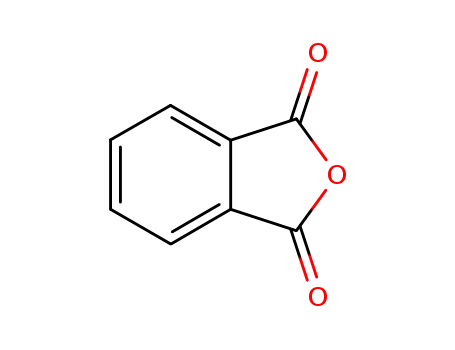
phthalic anhydride
3891-07-4 Downstream products
-
4537-76-2
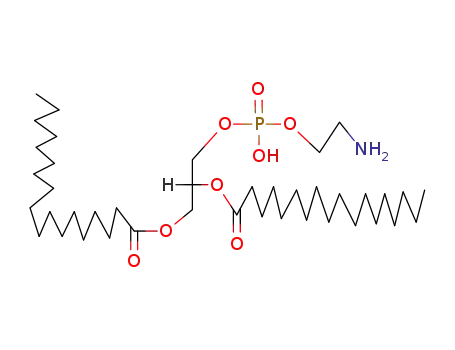
distearylphosphatidylethanolamine
-
20255-95-2
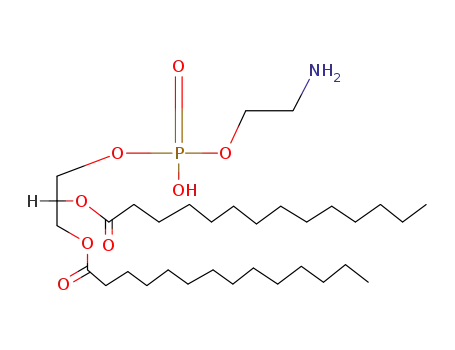
dimyristoylphosphatidylethanolamine
-
5466-90-0
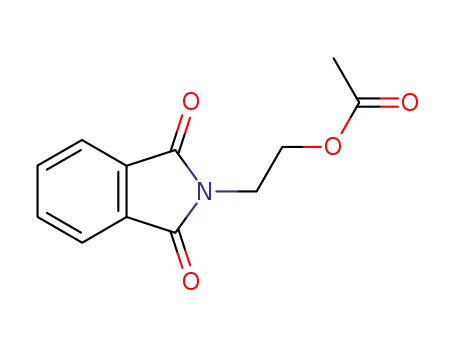
2-(1,3(2H)-dioxo-1H-isoindol-2-yl)ethyl acetate
-
6656-84-4
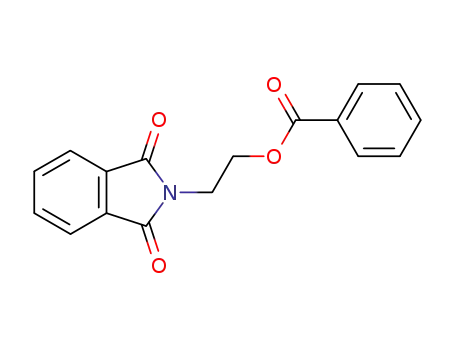
2-(1,3-dioxo-1,3-dihydro-2H-isoindol-2-yl)ethyl benzoate