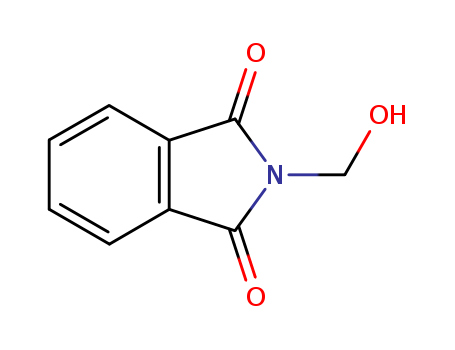


CasNo: 118-29-6
MF: C9H7NO3
Appearance: white crystalline powder
|
Definition |
ChEBI: A primary alcohol comprising phthalimide carrying an N-hydroxymethyl substituent. |
|
Preparation |
To a flask equipped with a mechanical stirrer and condenser is added 511 gm (3.47 moles) of phthalimide, 260 ml of 40% formalin (3.47 moles), and 1750 ml of water. The mixture is refluxed for about 5-10 min or until a clear solution results (if any insoluble material remains, it is first filtered). Then the mixture is cooled for several hours. The resulting product is filtered with suction, washed with cold water, and air-dried to afford 594 gm (96%), m.p. 137-141°C. NOTE: The product should not be oven-dried since it decomposes with the loss of formaldehyde. Recrystallization of the product from ethanol affords 94% recovery of the original material with the same melting point range. |
|
Synthesis Reference(s) |
Journal of the American Chemical Society, 77, p. 1913, 1955 DOI: 10.1021/ja01612a068 |
InChI:InChI=1/C9H7NO3/c11-5-10-8(12)6-3-1-2-4-7(6)9(10)13/h1-4,11H,5H2
A series of novel thiazolidine-2,4-dione...
A convenient and high yield method for t...
-
Multimodal imaging probes have attracted...
-
-
New 1,3-bis-N-acyliminium species genera...
-
The synthesis of soft alkyl phenolic eth...
N-Hydroxymethylphthalimide (HMPhI) ester...
Cathodic reduction of phthalimide anion ...
The invention discloses a method for syn...
Inhibitors targeting the epidermal growt...
The invention discloses a synthesis meth...
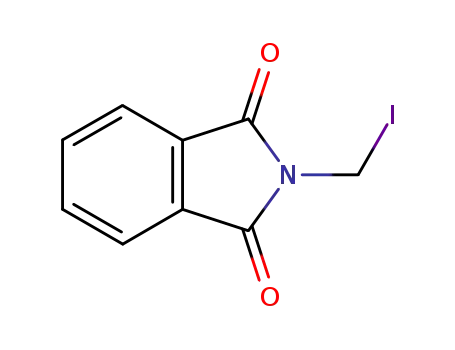
2-(iodomethyl)isoindoline-1,3-dione

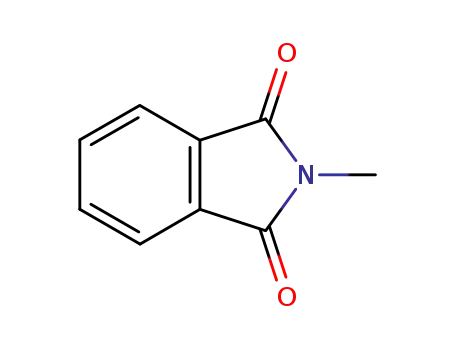
N-methylphthalimide

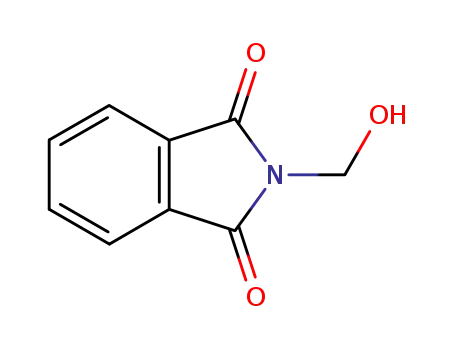
2-(hydroxymethyl)-1H-isoindole-1,3(2H)-dione

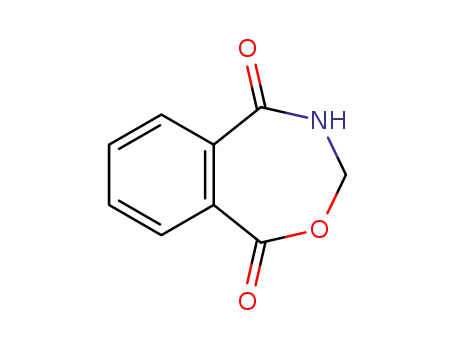
1,3,4,5-tetrahydro-2,4-benzoxazepine-1,5-dione
| Conditions | Yield |
|---|---|
|
With diethylzinc; In hexane; dichloromethane; at 20 ℃; for 2h; Inert atmosphere;
|
39% 14% 22% |
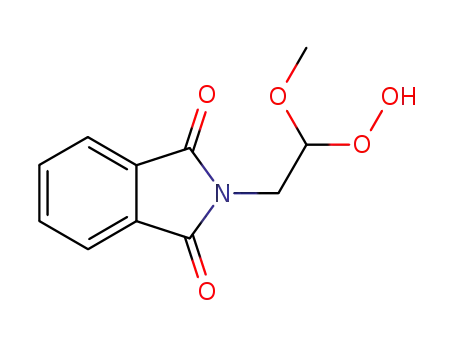
2-(2-Hydroperoxy-2-methoxy-ethyl)-isoindole-1,3-dione


N-methylphthalimide


2-(hydroxymethyl)-1H-isoindole-1,3(2H)-dione

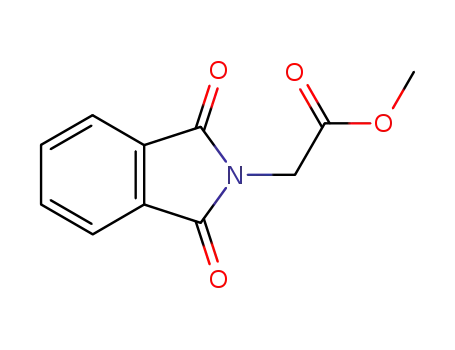
N-phthaloylglycine methyl ester
| Conditions | Yield |
|---|---|
|
In dimethyl sulfoxide; Irradiation;
|
35% 40% 15% |
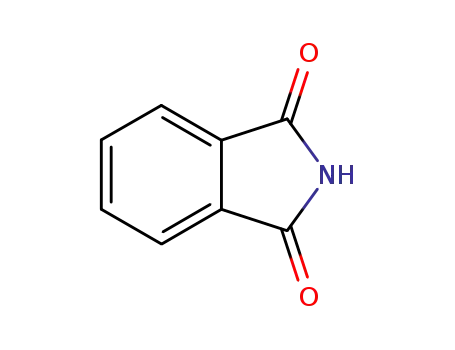
phthalimide
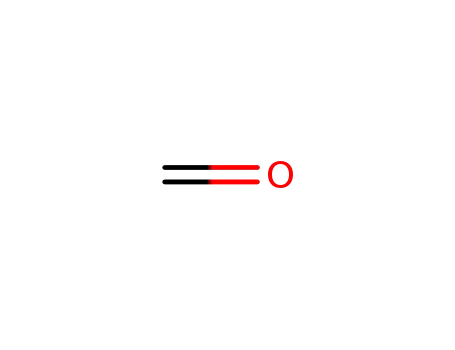
formaldehyd
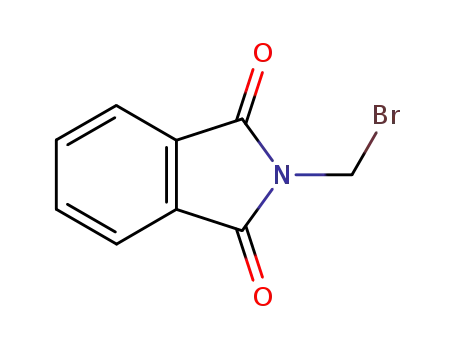
N-(bromomethyl)phtalimide
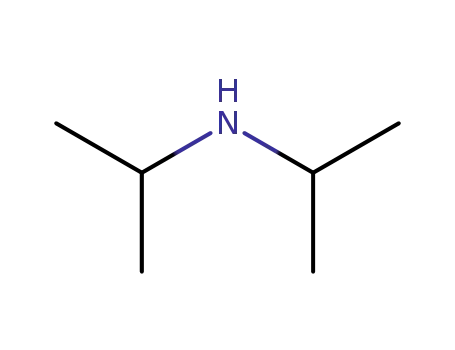
diisopropylamine
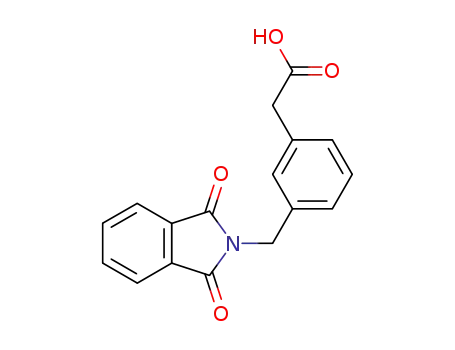
(3-phthalimidomethyl-phenyl)-acetic acid
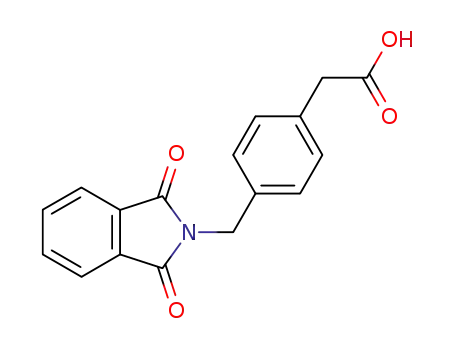
(4-phthalimidomethyl-phenyl)-acetic acid
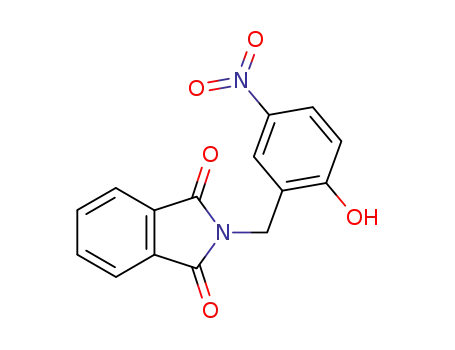
N-(2-hydroxy-5-nitro-benzyl)-phthalimide
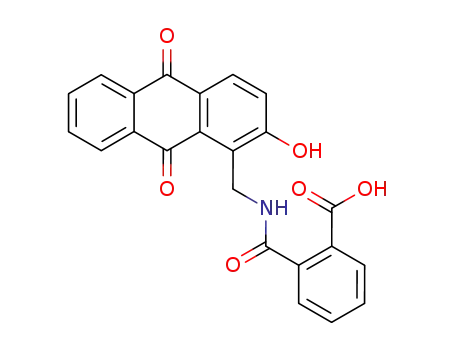
N-(2-hydroxy-9,10-dioxo-9,10-dihydro-[1]anthrylmethyl)-phthalamic acid