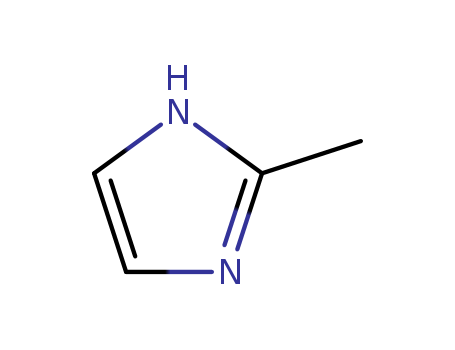


CasNo: 693-98-1
MF: C4H6N2
Appearance: white to light yellow crystalline powder
|
Preparation |
2-Methylimidazole is prepared by condensation of glyoxal, ammonia and acetaldehyde, a Radziszewski reaction. It is widely used as a polymeriza tion cross-linking accelerator and a hardener for epoxy resin systems for semiconductor potting compounds and soldering masks. It is obtained by eliminating dehydrogenation of 2-methylimidazoline. 2-methylimidazoline heated to melt (melting point 107 ℃), carefully add active nickel, raise the temperature to 200-210 ℃ reaction 2h. cool down to below 150 ℃, add water to dissolve, while hot pressure filtration, separation of active nickel, the filter lookchem liquid concentrated to a temperature of 140 ℃ or more, put the material cooling that 2-methylimidazole. Use the method to produce purity of ≥ 98% of the product, 1t product consumption of ethylenediamine (95%) 1095kg, acetonitrile 975kg. better method is to use glyoxal and aldehyde as raw materials. |
|
Hazard |
Possible carcinogen. |
|
Flammability and Explosibility |
Notclassified |
|
Purification Methods |
Recrystallise 2-methylimidazole from *benzene or pet ether. The picrate has m 215o (from H2O). [Beilstein 23 III/IV 594, 23/5 V 35.] |
|
General Description |
It is a white or colorless solid that is highly soluble in polar organic solvents and water. It is a precursor to a range of drugs and is a ligand in coordination chemistry. |
InChI:InChI=1/C4H6N2/c1-4-5-2-3-6-4/h2-3H,1H3,(H,5,6)
In situ formation of 2-methylimidazole a...
The invention provides a continuous synt...
Sophisticated chemical processes widely ...
The invention relates to a novel nitroim...
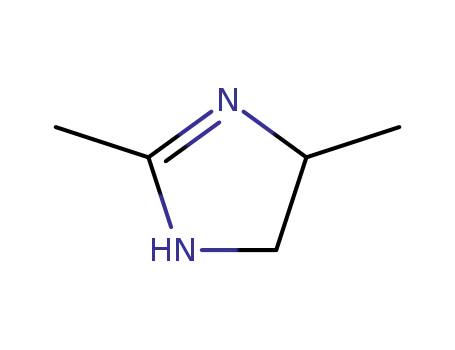
2,4-dimethyl-2-imidazoline

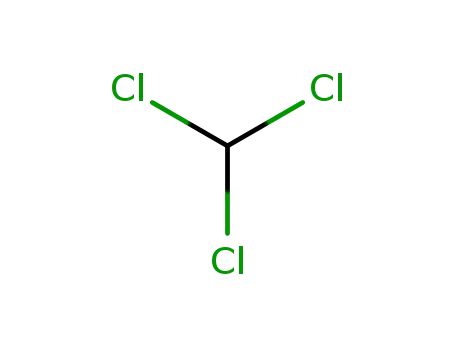
chloroform

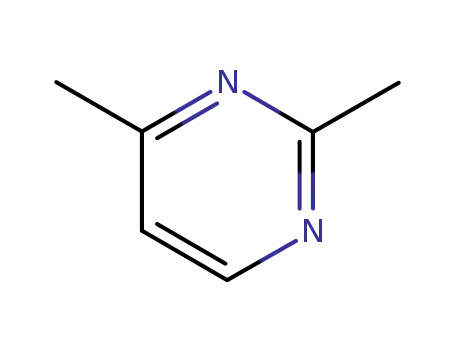
2,4-dimethylpyrimidine

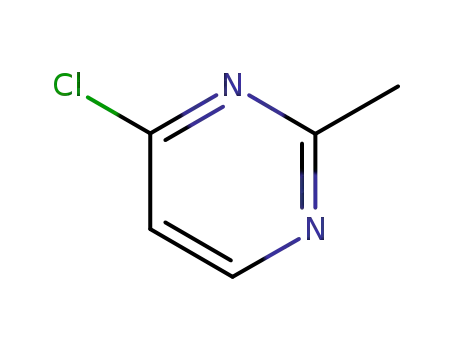
4-chloro-2-methylpyrimidine

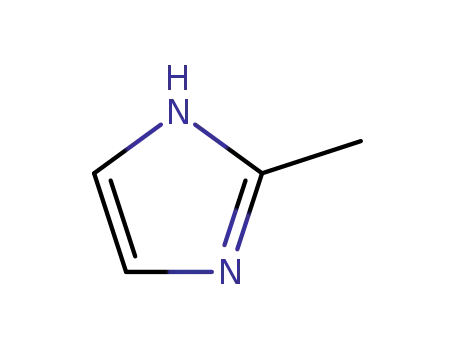
2-methylimidazole

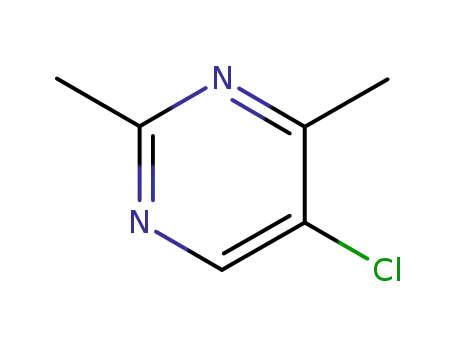
5-Chloro-2,4-dimethylpyrimidine
| Conditions | Yield |
|---|---|
|
at 550 ℃; Yield given. Further byproducts given. Yields of byproduct given. Title compound not separated from byproducts;
|

2,4-dimethyl-2-imidazoline


chloroform

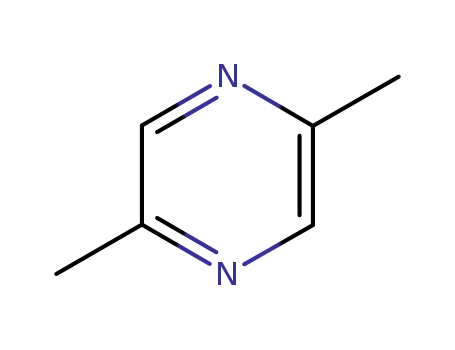
2,5-dimethyl-pyrazine


4-chloro-2-methylpyrimidine


2-methylimidazole


5-Chloro-2,4-dimethylpyrimidine
| Conditions | Yield |
|---|---|
|
at 550 ℃; Yield given. Further byproducts given. Yields of byproduct given. Title compound not separated from byproducts;
|
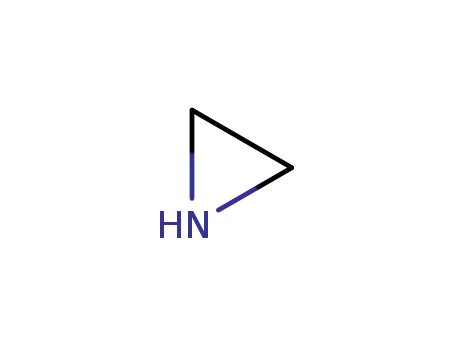
ethyleneimine
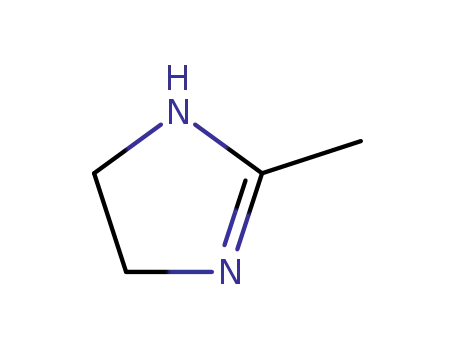
lysidine
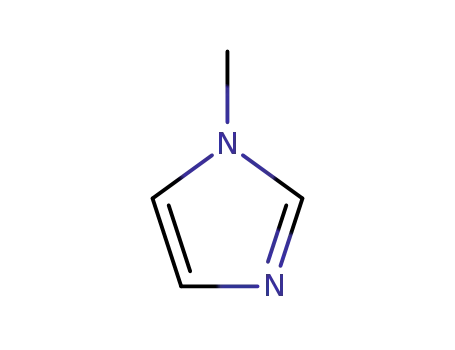
1-methyl-1H-imidazole
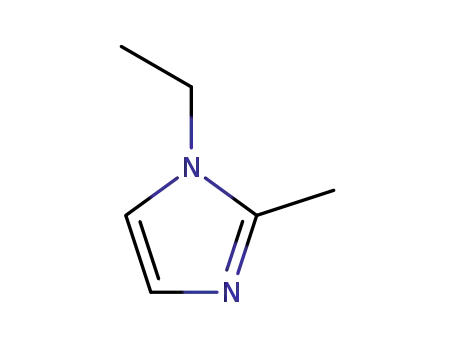
1-ethyl-2-methylimidazole
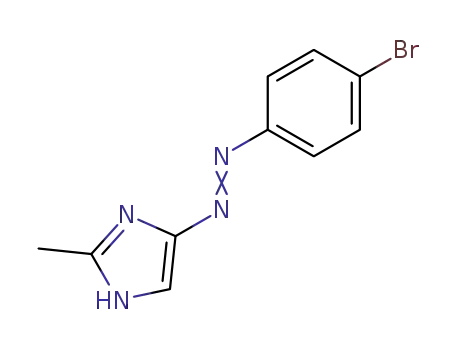
2-methyl-4-(p-bromophenylazo)-1H-imidazole
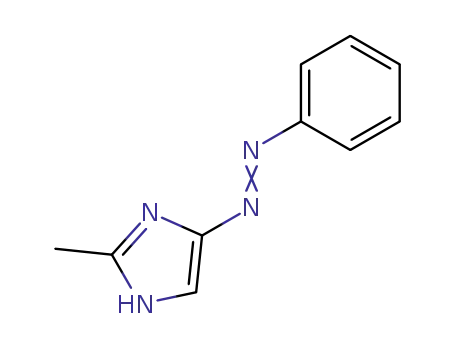
2-methyl-4-(phenylazo)-1H-imidazole
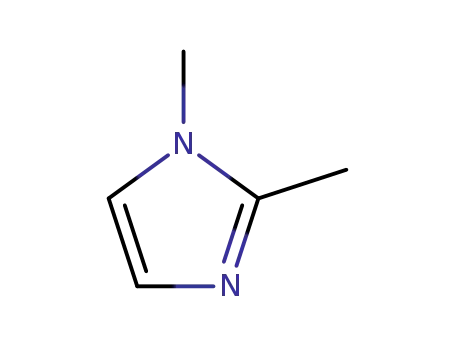
1,2-dimethyl-1H-imidazole
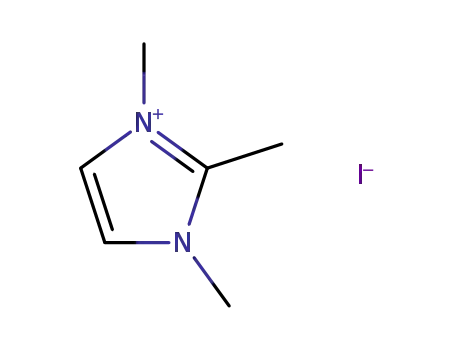
1,2,3-trimethylimidazolium iodide