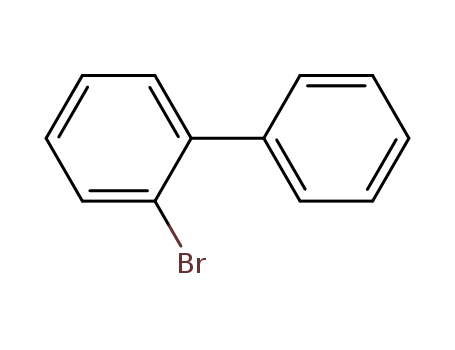
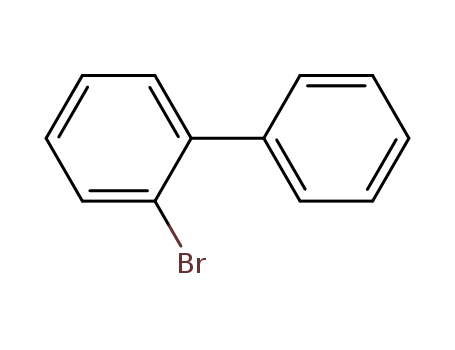
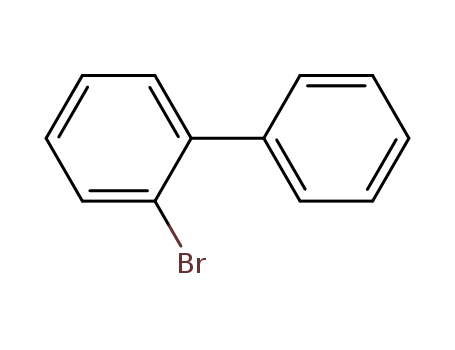
CasNo: 2052-07-5
MF: C12H9Br
Appearance: colourless liquid
|
synthesis |
Carry out all catalytic reactions in reaction vessels open to the air. Charge a round-bottom flask with the newly purchased or freshly recrystallized aryl boronic acid (2.0 mmol), 1,2-dibromobenzene, powdered K3PO4 (2.2 mmol) and toluene (2.5 mL) of technical quality. Stir the mixture vigorously. Heat the mixture to 80 °C for 10 minutes. Add (0.2 mol%) of catalyst by syringe as a 2.5 ml of toluene solution to the mixture. Take the samples periodically from the reaction mixture. Quench the reaction with water. Extract the mixture with ethyl acetate. Analyze the reaction by GC-MS. At the end of catalytic reaction, cool the reaction mixture to room temperature. Quench the mixture with water (adjusted to an appropriate pH when biaryls with acidic or basic groups has to be extracted). Extract the mixture with ethyl acetate (3×40 mL). Dry the combined extracts (MgSO4). Evaporate the combined extracts to dryness. Purify the crude material by flash chromatography on silica gel. |
|
Synthesis Reference(s) |
The Journal of Organic Chemistry, 44, p. 3037, 1979 DOI: 10.1021/jo01331a016Tetrahedron Letters, 21, p. 845, 1980 DOI: 10.1016/S0040-4039(00)71521-8 |
|
Air & Water Reactions |
Insoluble in water. |
|
Reactivity Profile |
2-Bromobiphenyl is sensitive to light. Low reactivity. |
|
Fire Hazard |
Flash point data for 2-Bromobiphenyl are not available. 2-Bromobiphenyl is probably combustible. |
|
Physical properties |
2-Bromobiphenyl has a melting point of 1.5-2 °C, so it is a colorless liquid at room temperature. It boils at 297-298 °C. Its density at 25℃ is 1.352 g/mL. In general, 2-bromobiphenyls are stable, but they are not compatible with oxidants. |
|
General Description |
Clear liquid. Insoluble in water. |
InChI:InChI=1/C12H9Br/c13-12-9-5-4-8-11(12)10-6-2-1-3-7-10/h1-9H
A new family of ferrocenylated P^N ligan...
The first magnetic nanocatalysts for C-H...
-
Donor-acceptor nanofibril composites hav...
A series of phosphonium salts with steri...
A new relay C–H functionalization of di(...
A novel electrocatalyzed method for the ...
Rhodium(III)-catalyzed annulation of 2-b...
We present the first synthesis of air/mo...
bromobenzene

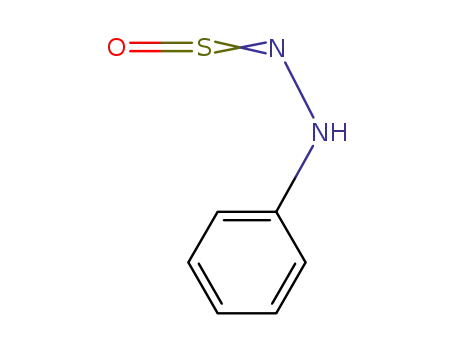
N-sulphinylphenylhydrazine

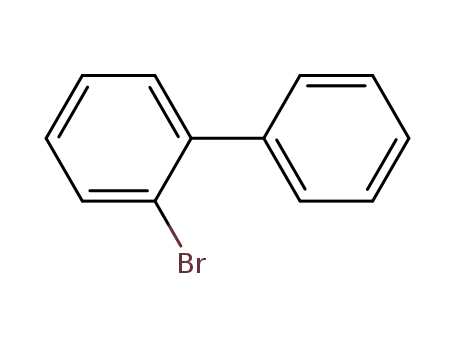
2-Bromobiphenyl

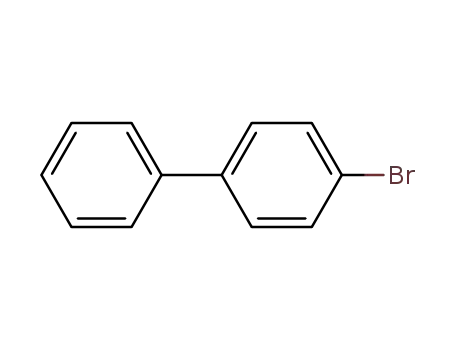
4-bromo-1,1'-biphenyl

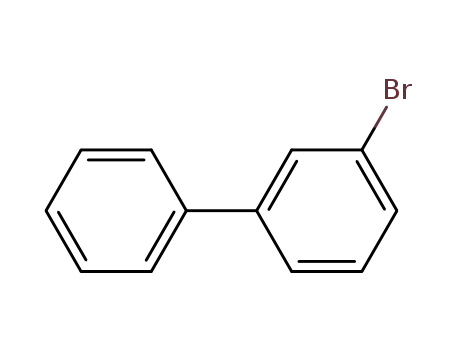
3-bromobiphenyl

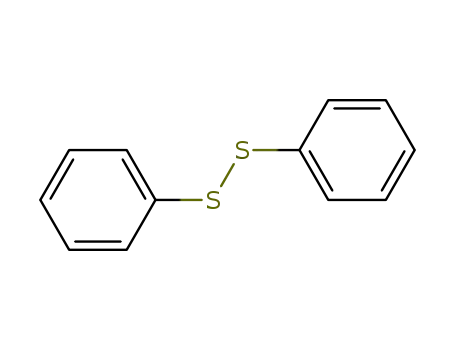
diphenyldisulfane
| Conditions | Yield |
|---|---|
|
at 135 ℃; for 72h; Further byproducts given;
|
12.7% 6.9% 20.9% 1.1% |
|
at 135 ℃; for 72h; Further byproducts given;
|
19.5% 12.9% 1.1% 8.1% |
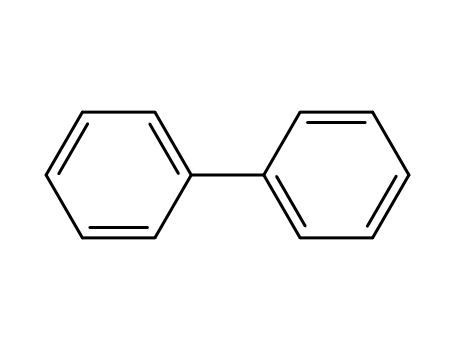
biphenyl

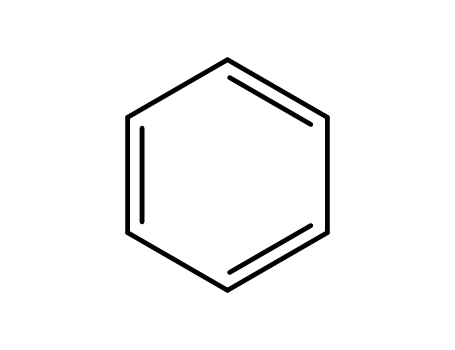
benzene

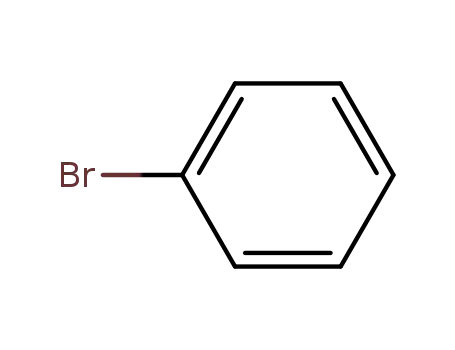
bromobenzene

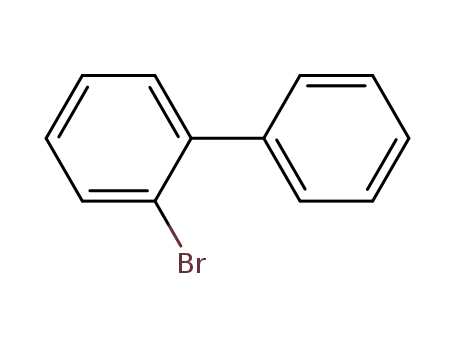
2-Bromobiphenyl

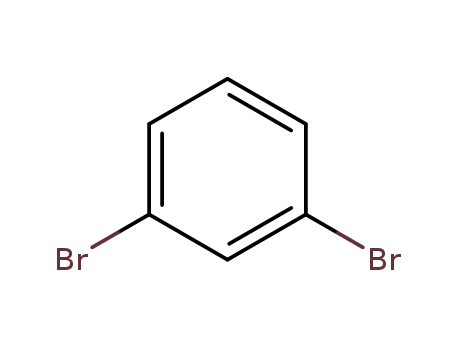
1,3-dibromobenzene

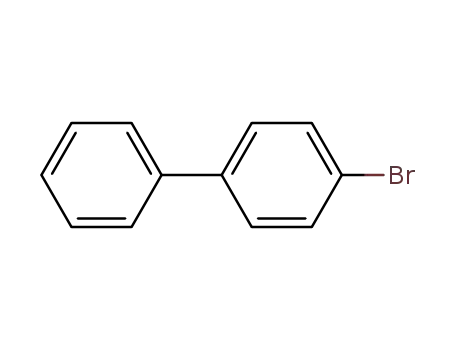
4-bromo-1,1'-biphenyl


3-bromobiphenyl

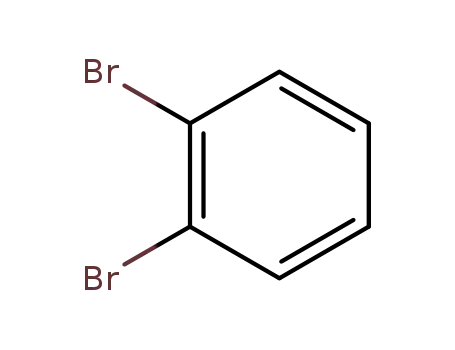
2,3-dibromobenzene
| Conditions | Yield |
|---|---|
|
With bromine; at 50 ℃; for 20h; Product distribution; Irradiation; other time, reagents ratio;
|
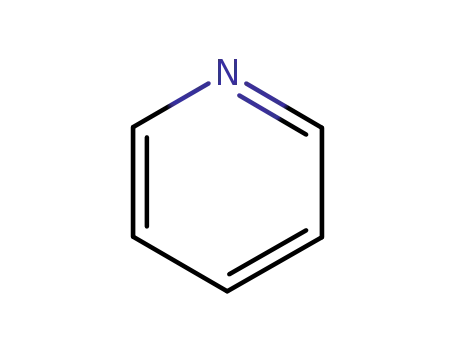
pyridine
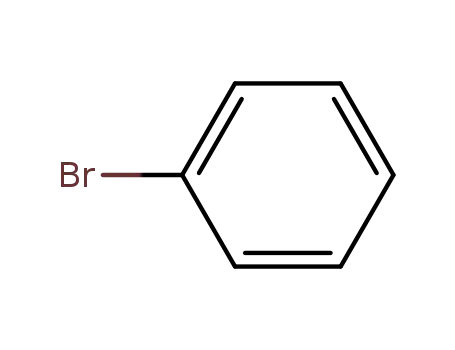
bromobenzene
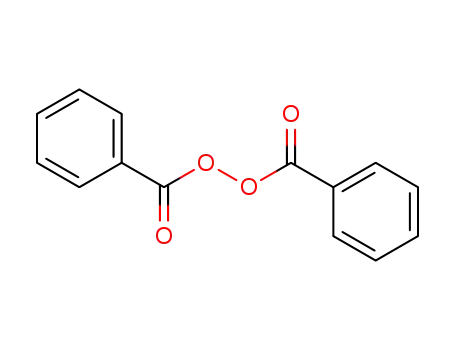
dibenzoyl peroxide

phosgene

2-phenylbenzophenone
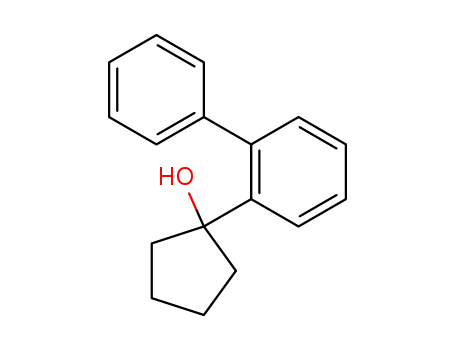
1-biphenyl-2-yl-cyclopentanol
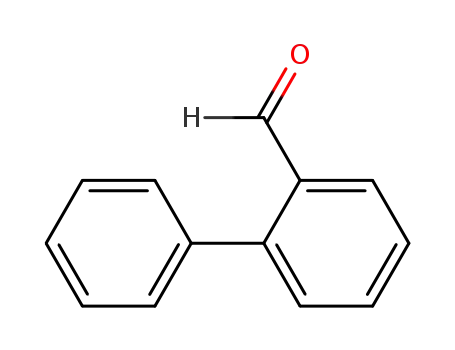
2-Phenylbenzaldehyde
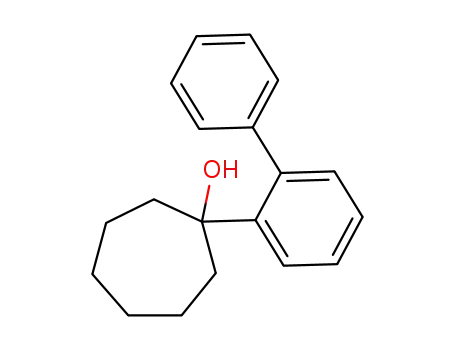
1-Biphenyl-2-yl-cycloheptanol