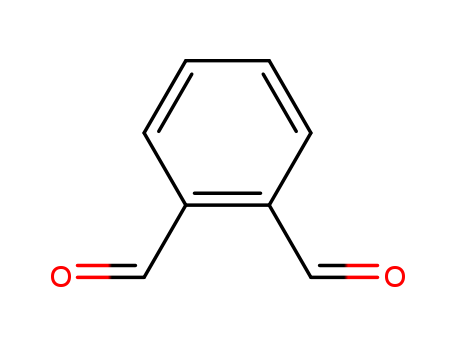


CasNo: 643-79-8
MF: C8H6O2
Appearance: Light yellow powder
|
Chemical Description |
o-Phthalaldehyde is a chemical compound used in organic synthesis and as a fluorescent probe for amino acids. |
|
Preparation |
o-Phthalaldehyde is a high-level chemical disinfectant that is commonly used for disinfection of dental and medical instruments as an alternative to glutaraldehyde, which is a known skin and respiratory sensitizer. A variety of processes for manufacturing o-phthalaldehyde have been reported in the literature. o-Phthalaldehyde is produced by heating pure benzaldehyde and chloroform with potassium hydroxide solution. The resulting solution is further acidified with hydrochloric acid and cooled to yield a colorless powder of o-phthalaldehyde. It is also produced by ozonization of naphthalene in alcohol followed by catalytic hydrogenation. Catalytic oxidation of various chemicals is also used in manufacturing o-phthalaldehyde. o-Phthalaldehyde can be manufactured by oxidation of phthalan by nitrogen monoxide in acetonitrile with N-hydroxyphthalimide as the catalyst to yield 80% to 90%. |
|
Synthesis Reference(s) |
Journal of the American Chemical Society, 73, p. 1668, 1951 DOI: 10.1021/ja01148a076Tetrahedron Letters, 27, p. 1793, 1986 DOI: 10.1016/S0040-4039(00)84377-4 |
|
Flammability and Explosibility |
Notclassified |
|
Potential Exposure |
The primary routes of human exposure to o-phthalaldehyde are by inhalation and through the skin, which may occur through accidental or occupational exposures. Along with its increasing popularity as a chemical sterilizer, o-phthalaldehyde has many applications in analytical methods and in diagnostic kits. o-Phthalaldehyde is also used as an intermediate in the synthesis of pharmaceuticals and as a reagent in the tanning industry, hair colorings, wood treatment, and antifouling paints. o-Phthalaldehyde was approved for use as an indoor antimicrobial pesticide in 1997; however, it is no longer registered with the United States Environmental Protection Agency (USEPA) for this use. |
|
Carcinogenicity |
No information on the carcinogenicity of o-phthalaldehyde in experimental animals or humans was found in a review of the literature. |
|
Definition |
ChEBI: A dialdehyde in which two formyl groups are attached to adjacent carbon centres on a benzene ring. |
|
Biotechnological Applications |
O-phthalaldehyde(OPA) is used for precolumn derivatization of amino acids for HPLC separation and for flow cytometric measurements of protein thiol groups. Used for fluorometric determination of histamine, histidine and other amino acids. Also used for cholesterol assay in the picomole range. Phthaldialdehyde has been used: in the preparation of O-phthaldialdehyde reagent for analysing gentamycin content. in the preparation of reagent for determining the degree of hydrolysis of milk proteins. in the measurement of free amino acids of milk samples by O-phthaldialdehyde/N-acetyl-L-cysteine (OPA/NAC) assay. in the derivatization of putrescine samples. |
InChI:InChI=1/C8H6O2/c9-5-7-3-1-2-4-8(7)6-10/h1-6H
The oxidation of ortho-phthalaldehyde to...
Room temperature and atmosphere pressure...
Synthesis of new Schiff's base Zn-comple...
A magnetically recyclable palladium-cata...
The reactivity of an electronically tune...
A new synthesis of phthalaldehyde that a...
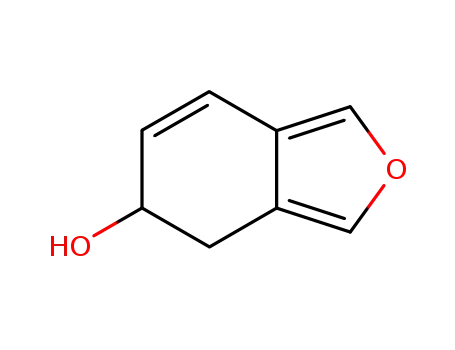
5-hydroxy-4,5-dihydroisobenzofuran

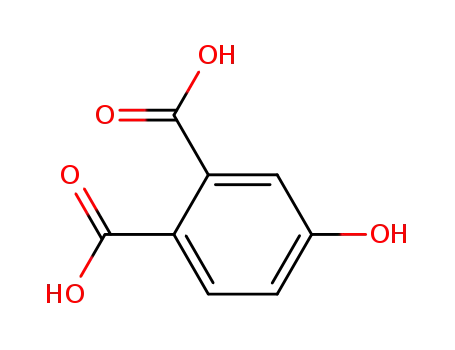
4-hydroxyphthalic acid

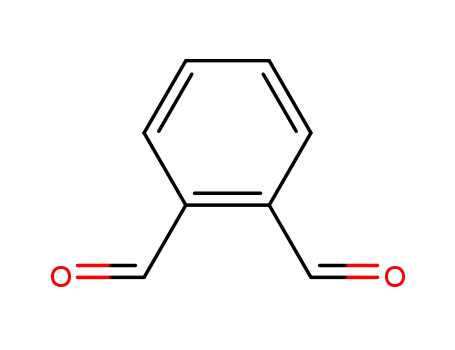
o-phthalic dicarboxaldehyde
| Conditions | Yield |
|---|---|
|
With selenium(IV) oxide; In 1,4-dioxane;
|
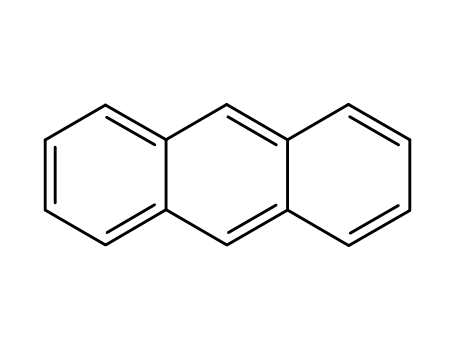
anthracene

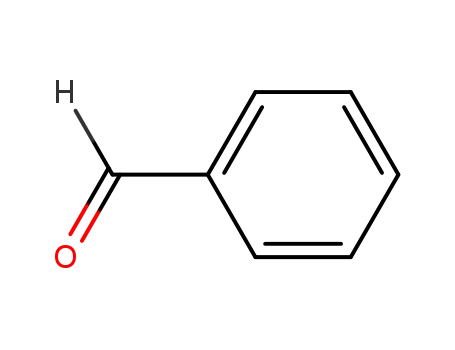
benzaldehyde

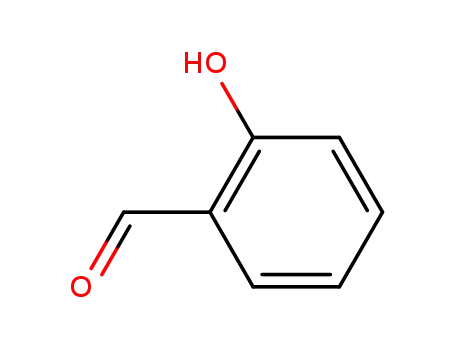
salicylaldehyde

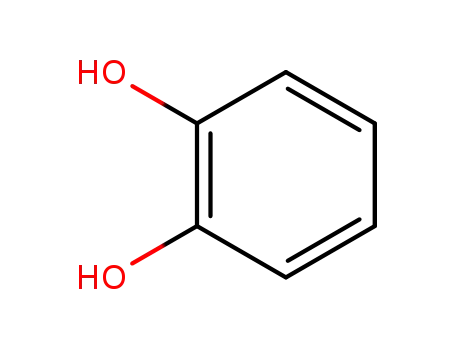
benzene-1,2-diol


o-phthalic dicarboxaldehyde
| Conditions | Yield |
|---|---|
|
In water; Further byproducts given; Formation of xenobiotics; simulated solar irradiation;
|
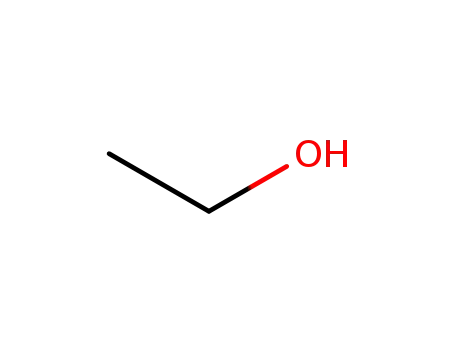
ethanol
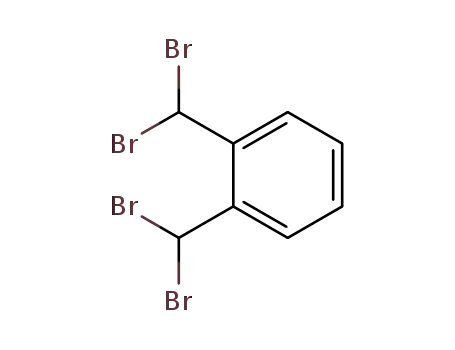
1,2-bis(dibromomethyl)benzene
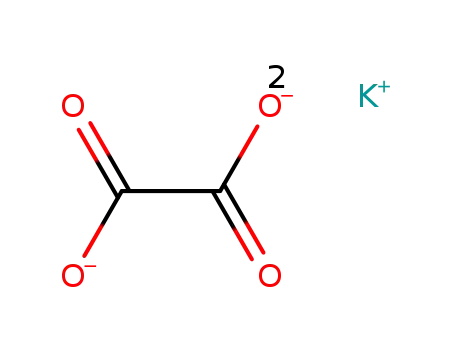
potassium oxalate
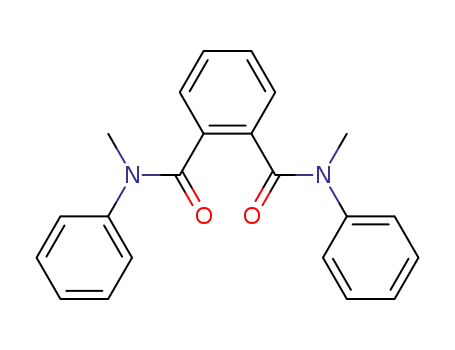
N,N'-dimethyl-N,N'-diphenyl-phthalamide
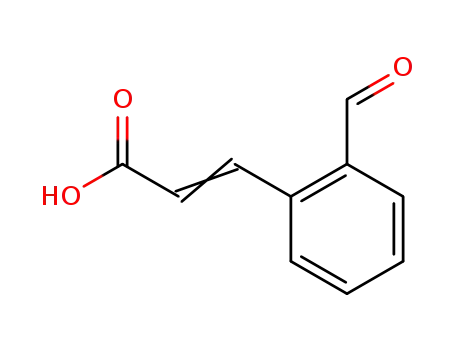
2-Formylcinnamic acid
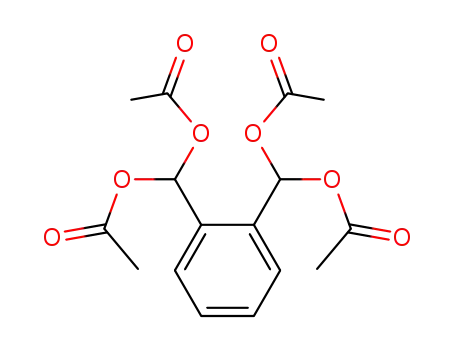
1,2-bis-diacetoxymethyl-benzene
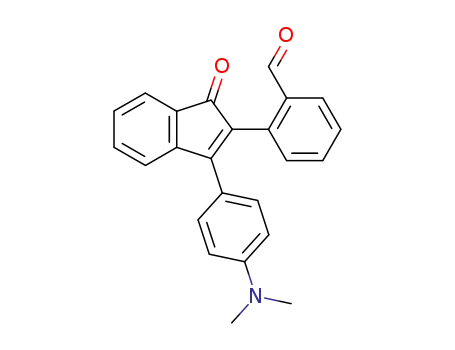
2-[3-(4-dimethylamino-phenyl)-1-oxo-inden-2-yl]-benzaldehyde

2,7-Nonamethylen-4,5-benzotropon