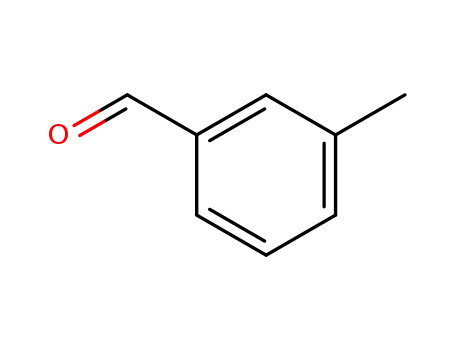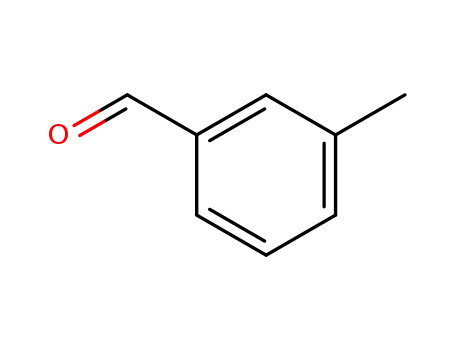Quality Manufacturer Supply Wholesale 626-19-7 with Reasonable Price
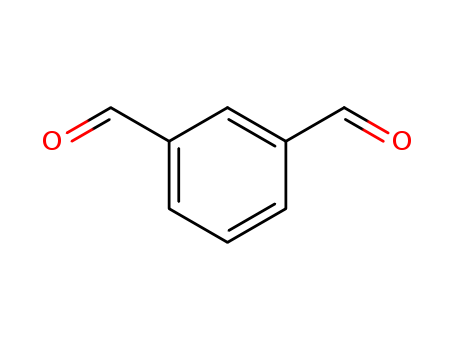
- Molecular Formula:C8H6O2
- Molecular Weight:134.134
- Appearance/Colour:colourless or light yellow crystals
- Vapor Pressure:0.0164mmHg at 25°C
- Melting Point:87-88 °C(lit.)
- Refractive Index:1.622
- Boiling Point:255.3 °C at 760 mmHg
- Flash Point:94.1 °C
- PSA:34.14000
- Density:1.189 g/cm3
- LogP:1.31160
m-Phthalaldehyde(Cas 626-19-7) Usage
|
Synthesis Reference(s)
|
Tetrahedron Letters, 36, p. 455, 1995 DOI: 10.1016/0040-4039(94)02284-I
|
|
General Description
|
Isophthalaldehyde participates in base-catalyzed Knoevenagel condensation reaction.
|
InChI:InChI=1/C8H6O2/c9-5-7-2-1-3-8(4-7)6-10/h1-6H
626-19-7 Relevant articles
A Magnetically Recyclable Palladium-Catalyzed Formylation of Aryl Iodides with Formic Acid as CO Source: A Practical Access to Aromatic Aldehydes
You, Shengyong,Zhang, Rongli,Cai, Mingzhong
supporting information, p. 1962 - 1970 (2021/01/25)
A magnetically recyclable palladium-cata...
Preparation method for synthesizing aryl aldehyde compounds by reducing aryl secondary amide or aryl secondary amide derivative through phenylsilane
-
Paragraph 0062-0066, (2021/11/10)
The invention provides a preparation met...
Synthesis of new Zn (II) complexes for photo decomposition of organic dye pollutants, industrial wastewater and photo-oxidation of methyl arenes under visible-light
Ahemed, Jakeer,Bhongiri, Yadagiri,Chetti, Prabhakar,Gade, Ramesh,Kore, Ranjith,Pasha, Jakeer,Pola, Someshwar,Rao D, Venkateshwar
, (2021/07/28)
Synthesis of new Schiff's base Zn-comple...
A Remote ‘Imidazole’-Based Ruthenium(II) Para-Cymene Pre-catalyst for the Selective Oxidation Reaction of Alkyl Arenes and Alcohols
Dutta, Manali,Bania, Kusum K.,Pratihar, Sanjay
, p. 926 - 932 (2020/03/05)
Herein we disclosed the use of a remote ...
626-19-7 Process route
-
- 108-38-3,104809-90-7
m-xylene
-
- 626-19-7
Isophthalaldehyde
-
- 620-14-4
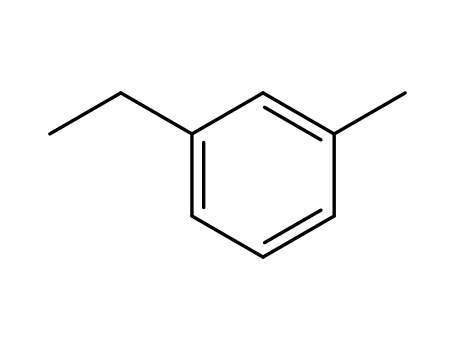
1-Methyl-3-ethylbenzene
-
- 4662-96-8
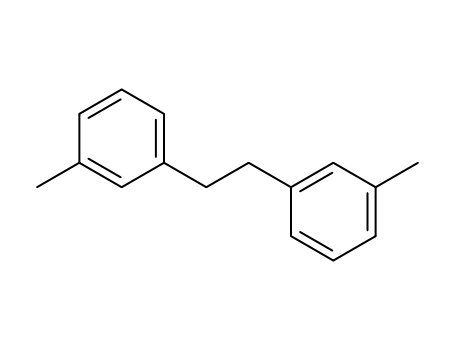
1,2-di-m-tolylethane
-
- 108-88-3,15644-74-3,16713-13-6
toluene
-
- 620-23-5
m-tolyl aldehyde
-
- 71-43-2,26181-88-4,54682-86-9,13967-78-7,174973-66-1
benzene
Conditions
| Conditions |
Yield |
|
With oxygen; at 881.9 ℃; under 760 Torr; Mechanism; Product distribution;
|
|
-
- 108-38-3,104809-90-7
m-xylene
-
- 626-19-7
Isophthalaldehyde
-
- 620-23-5
m-tolyl aldehyde
Conditions
| Conditions |
Yield |
|
With oxygen; 10-methyl-9-phenylacridin-10-ium perchlorate; In chloroform; at 24.84 ℃; for 10h; Title compound not separated from byproducts.; Pyrolysis; visible light;
|
|
626-19-7 Upstream products
-
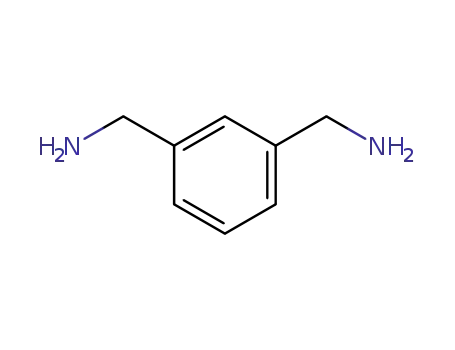
1477-55-0
1,3-di(aminomethyl)benzene
-
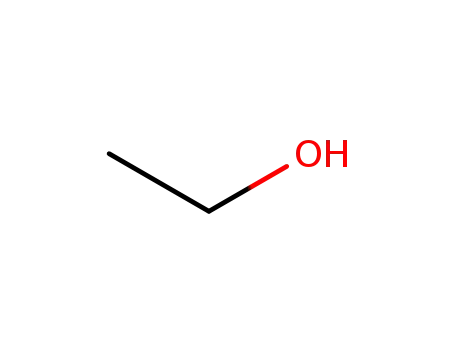
64-17-5
ethanol
-
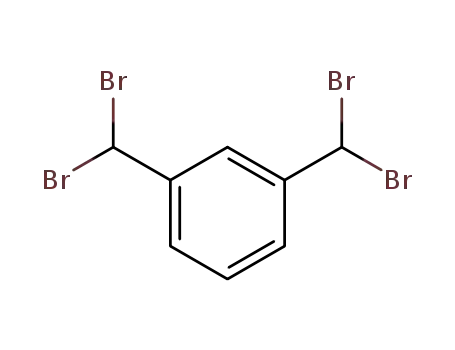
36323-28-1
α,α,α',α'-tetrabromo-m-xylene
-
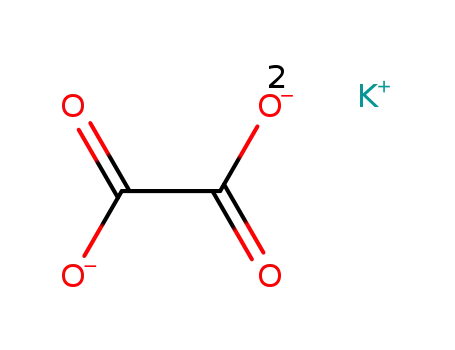
583-52-8
potassium oxalate
626-19-7 Downstream products
-
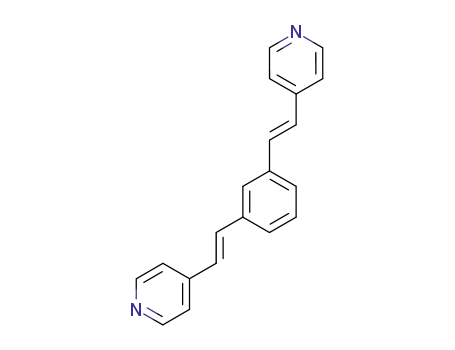
21915-02-6
1,3-bis((E)-2-(pyridin-4-yl)vinyl)benzene
-
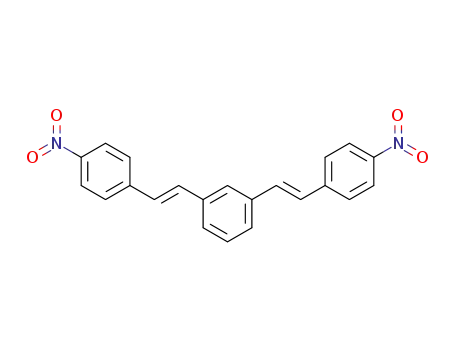
2695-65-0
1,3-bis-(4-nitro-trans-styryl)-benzene
-
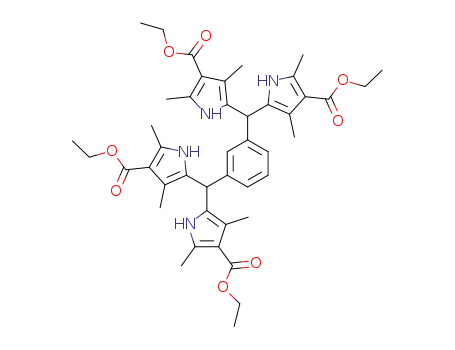
120613-22-1
1,3-bis-[bis-(4-ethoxycarbonyl-3,5-dimethyl-pyrrol-2-yl)-methyl]-benzene
-
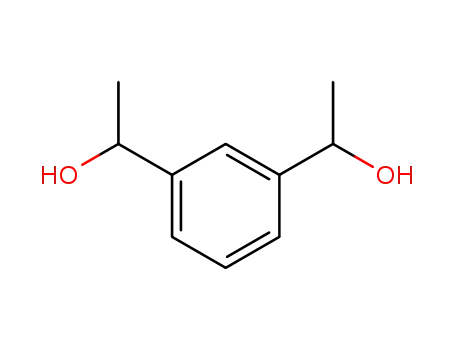
143329-81-1
α,α'-dimethyl-1,3-benzenedimethanol