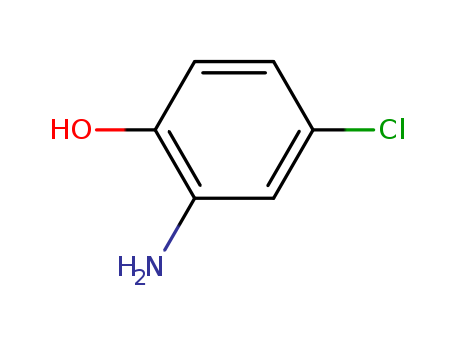


CasNo: 95-85-2
MF: C6H6ClNO
Appearance: brown crystalline solid
|
Air & Water Reactions |
5-Chloro-2-hydroxyaniline may be sensitive to prolonged exposure to air and/or light. Insoluble in water. |
|
Reactivity Profile |
5-Chloro-2-hydroxyaniline may react with strong oxidizing agents. |
|
Health Hazard |
Highly toxic, may be fatal if inhaled, swallowed or absorbed through skin. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution. |
|
Fire Hazard |
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Containers may explode when heated. Runoff may pollute waterways. |
|
Safety Profile |
A poison. Moderately toxic by ingestion. Mutation data reported. When heated to decomposition it emits very toxic fumes of Cland NOx. See also AROMATIC MINES and CHLORIDES. |
|
Potential Exposure |
Used as a chemical raw material, especially in dye manufacture |
|
Shipping |
UN2673 2-Amino-4-chlorophenol, Hazard Class: 6.1; Labels: 6.1-Poisonous materials |
|
Incompatibilities |
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides, iron, moisture and temperatures .43 C. |
|
General Description |
5-Chloro-2-hydroxyaniline is a light brown colored crystalline solid. 5-Chloro-2-hydroxyaniline may be toxic by ingestion. 5-Chloro-2-hydroxyaniline is insoluble in water. 5-Chloro-2-hydroxyaniline is used to make other chemicals. |
InChI:InChI=1/C6H6ClNO/c7-4-1-2-6(9)5(8)3-4/h1-3,9H,8H2
Process parameters were studied to incre...
2-Amino-4-chlorophenol was found to be t...
Herein, a novel designed heterogeneous c...
Herein, silver nanoparticles (Ag NPs), a...
Herein, we present an organic–inorganic ...
A series of 27 compounds of general stru...
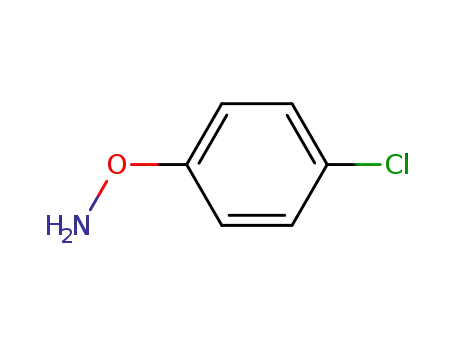
O-(4-chlorophenyl)hydroxylamine

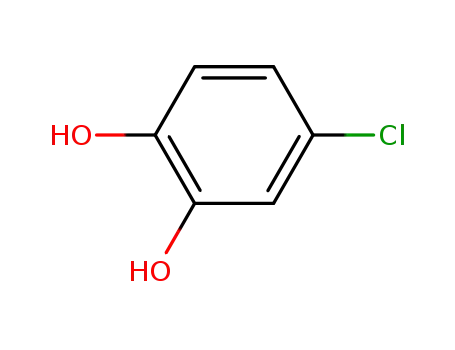
4-chloro-1,2-benzenediol

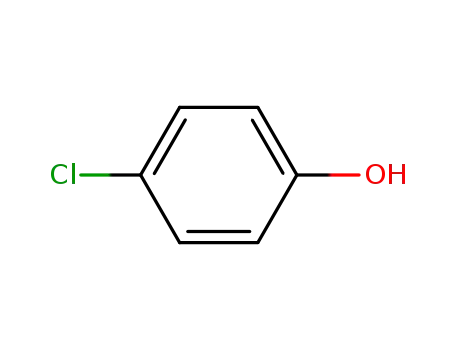
4-chloro-phenol

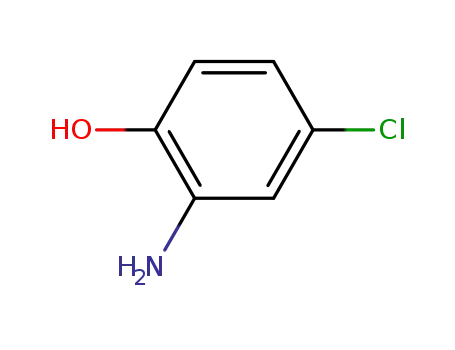
2-hydroxy-5-chloro-aniline
| Conditions | Yield |
|---|---|
|
With trifluoroacetic acid; at 30 ℃; for 5.5h; Product distribution; Rate constant; Kinetics; ΔH(excit.), ΔG(excit.), ΔS(excit.);
|
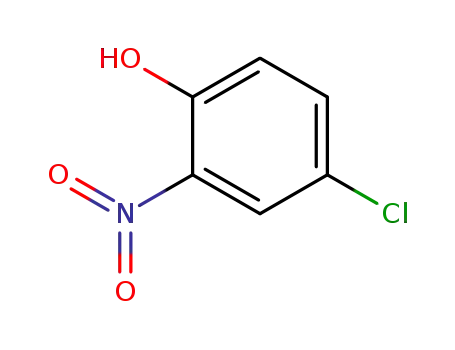
p-chloro-o-nitrophenol


2-hydroxy-5-chloro-aniline
| Conditions | Yield |
|---|---|
|
With hydrazine hydrate; In water; at 110 ℃; Sealed tube; Green chemistry;
|
99% |
|
With hydrazine hydrate; In ethanol; at 70 ℃; for 0.133333h;
|
96% |
|
With sodium tetrahydroborate; water; potassium carbonate; at 70 ℃; for 7h; pH=Ca. 8; Green chemistry;
|
96% |
|
With hydrazine hydrate; In isopropyl alcohol; at 110 ℃; for 0.2h; chemoselective reaction; Catalytic behavior; Sealed tube;
|
94% |
|
With hydrazine hydrate; In ethanol; at 70 ℃; for 0.116667h;
|
92% |
|
With (phthalocyaninato)iron(II); diphenylsilane; In ethanol; at 100 ℃; for 24h; Reagent/catalyst;
|
86% |
|
With tin(IV) chloride; pyrographite; hydrazine hydrate; In methanol; at 70 ℃; for 14h; Temperature; Reagent/catalyst; Large scale;
|
86.3% |
|
With ethanol; palladium on activated charcoal; sodium hydroxide; silicon; at 20 ℃; for 72h; Schlenk technique;
|
86% |
|
With copper(II) phthalocyanine; hydrazine hydrate; In ethylene glycol; at 120 ℃; for 2h; chemoselective reaction;
|
80% |
|
With sulfur; aluminum oxide; sodium hydroxide; at 80 ℃; for 3h;
|
69% |
|
p-chloro-o-nitrophenol; With sulfuric acid; In water; at 100 ℃; for 4.25h; pH=4.5;
With sodium carbonate; In water; at 100 ℃; for 1.25h; pH=7 - 12;
|
61% |
|
With ammonium chloride; zinc; In ethanol; water; for 0.416667h;
|
48% |
|
With sodium dithionite;
|
|
|
With nickel; Hydrogenation;
|
|
|
With sulfuric acid; at 70 ℃; durch elektrolytische Reduktion an einer Bleikathode;
|
|
|
With hydrogenchloride; iron;
|
|
|
With hydrogenchloride; tin;
|
|
|
With hydrazine hydrate; nickel; In ethanol;
|
|
|
With hydrogen; palladium on activated charcoal; In ethanol;
|
|
|
With hydrogen; platinum; 3-(2-hydroxyethyl)-1-methyl-1H-imidazol-3-ium tetrafluoroborate; at 89.84 ℃; for 2h; under 7500.75 Torr; Autoclave;
|
|
|
With C32H16FeN8*Fe(2+)*7H2O*O4S(2-); hydrazine hydrate; In ethanol; water; at 120 ℃; chemoselective reaction;
|
99 %Chromat. |
|
With hydrogen; platinum on carbon; In methanol; for 1h; under 2844.39 Torr;
|
|
|
p-chloro-o-nitrophenol; With sulfuric acid; In water; at 100 ℃; for 2h; pH=4.5;
With sodium carbonate; In water; at 100 ℃; for 0.5h; pH=12;
|
|
|
With 5%-palladium/activated carbon; hydrogen; In methanol; at 100 ℃; for 5h; under 19001.3 Torr; Concentration; Pressure; Reagent/catalyst; Solvent; Temperature; Kinetics; Autoclave;
|
|
|
With iron; acetic acid; In water; for 0.75h; Reflux;
|
|
|
With hydrogen; In methanol; at 20 ℃; under 750.075 - 1500.15 Torr; Autoclave;
|
7.4 g |
|
With iron; acetic acid; In ethanol; water;
|
|
|
With sodium tetrahydroborate; In water; at 20 ℃; for 0.0666667h; chemoselective reaction;
|
|
|
With hydrazine hydrate; In water; at 110 ℃; for 1h; Sealed tube;
|

p-chloro-o-nitrophenol
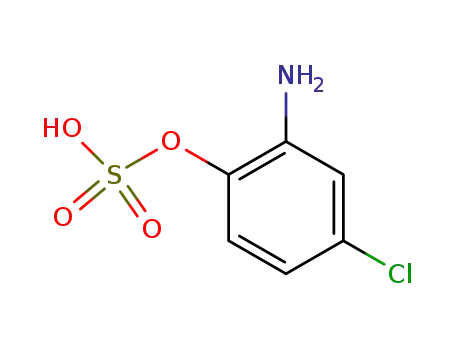
sulfuric acid mono-(2-amino-4-chloro-phenyl ester)
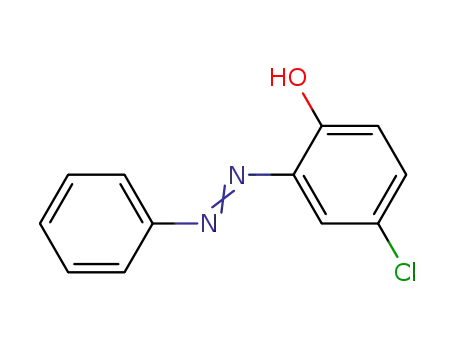
2-hydroxy-5-chloroazobenzene

O-(4-chlorophenyl)hydroxylamine
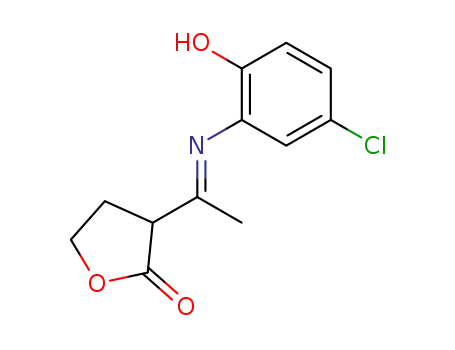
3-[1-(5-chloro-2-hydroxy-phenylimino)-ethyl]-dihydro-furan-2-one
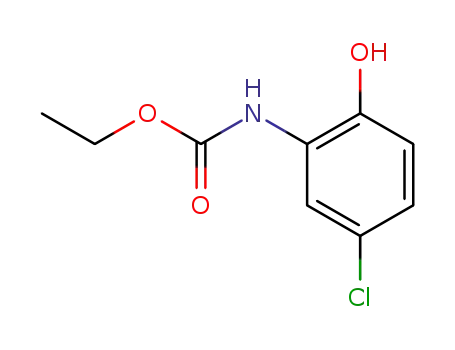
(5-chloro-2-hydroxy-phenyl)-carbamic acid ethyl ester
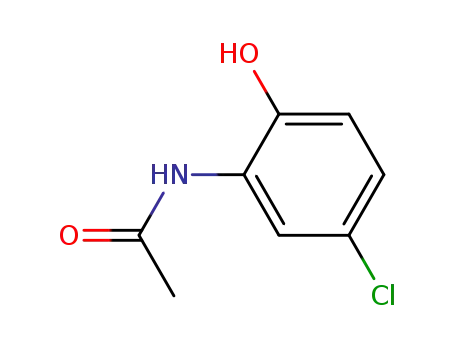
N-(5-chloro-2-hydroxyphenyl)acetamide
4-chloro-2-(4-nitro-benzylidenamino)-phenol