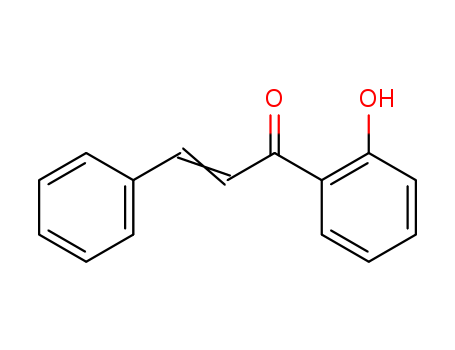
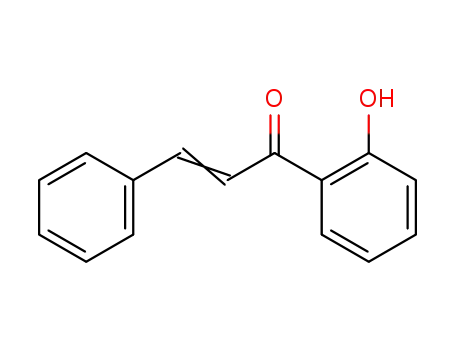
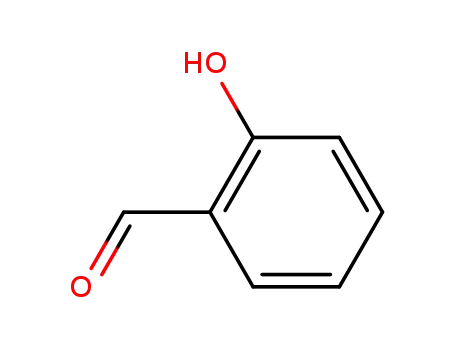
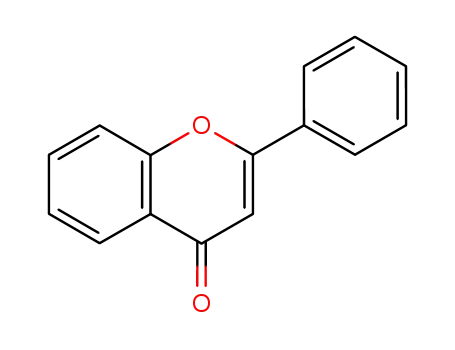
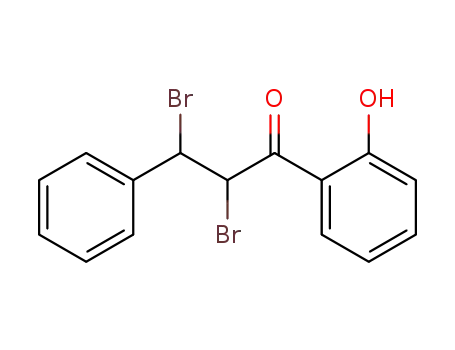
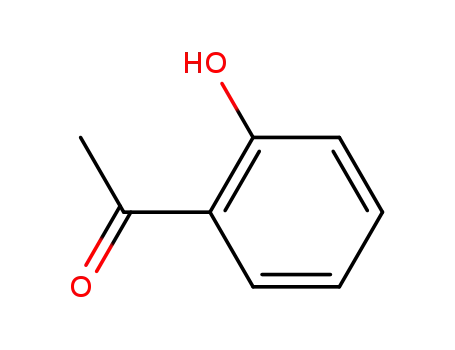
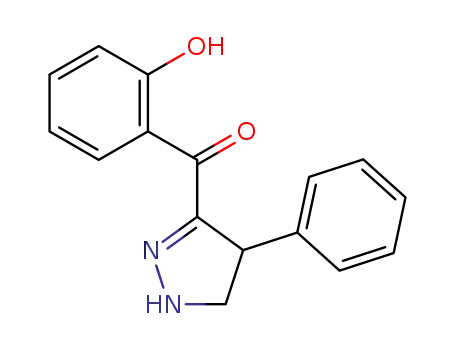
Low DNA and high BSA binding affinity of cationic ruthenium(II) organometallic featuring pyridine and 2’-hydroxychalcone ligands
Zahirovi?, Adnan,Roca, Sun?ica,Kahrovi?, Emira,Vi?njevac, Aleksandar
, (2021)
The chiral-at-metal, piano-stool rutheni...
Activated charcoal-mediated synthesis of chalcones catalyzed by NaOH in water
Tanemura, Kiyoshi,Rohand, Taoufik
, (2021)
A variety of chalcones were synthesized ...
Exploring the anti-breast cancer potential of flavonoid analogs
Thakor, Vanrajsinh,Poddar, Mayur,Dey, Sumit,Manjula,Madhunapantula, Subbarao V.,Pawara, Rahul,Patel, Harun M.,Noolvi, Malleshappa N.
, p. 79166 - 79179 (2016)
In the course of our search for new anti...
Cytotoxic activity of substituted chalcones in terms of molecular electronic properties
Kupcewicz, Bogumila,Jarzecki, Andrzej A.,Malecka, Magdalena,Krajewska, Urszula,Rozalski, Marek
, p. 4260 - 4265 (2014)
Global chemical reactivity descriptors a...
Synthesis of Hierarchically Porous Zeolite Composites with Enhanced Catalytic Activity: Effect of Different Long-Chain Structure Directing Agents
Ding, He,Zhang, Yixiao,Xiao, Zixing,Zhang, Jingshuang,Bai, Peng,Li, Najun,Guo, Xianghai
, p. 1730 - 1737 (2018)
A two-section temperature strategy was e...
Li-Al layered double hydroxides as catalysts for the synthesis of flavanone
French, Dustin,Schifano, Paul,Cortés-Concepción, José,Hargrove-Leak, Sirena
, p. 92 - 94 (2010)
Flavanone was synthesized via a series r...
Primary, secondary, and tertiary silanamine sites formed on nitrided SBA-15 for base catalytic C–C bond formation reactions
Moteki, Takahiko,Koga, Yuuki,Ogura, Masaru
, p. 131 - 139 (2019)
The relationship between catalyst prepar...
Modified Pyridine-Substituted Coumarins: A New Class of Antimicrobial and Antitubercular Agents
Giri, Rakesh R.,Lad, Hemali B.,Bhila, Varun G.,Patel, Chirag V.,Brahmbhatt
, p. 363 - 375 (2015)
Some new biologically potent coumarin de...
Synthesis of flavanones using nanocrystalline MgO
Choudary,Ranganath,Yadav, Jagajit,Lakshmi Kantam
, p. 1369 - 1371 (2005)
The design and development of a truly na...
A new ratiometric ESIPT sensor for detection of palladium species in aqueous solution
Liu, Bin,Wang, Hu,Wang, Taisheng,Bao, Yinyin,Du, Fanfan,Tian, Jiao,Li, Qianbiao,Bai, Ruke
, p. 2867 - 2869 (2012)
An aqueous ratiometric ESIPT sensor with...
Synthesis of nanocrystalline MFI-zeolites with intracrystal mesopores and their application in fine chemical synthesis involving large molecules
Srivastava, Rajendra,Iwasa, Nobuhiro,Fujita, Shin-Ichiro,Arai, Masahiko
, p. 9507 - 9511 (2008)
Synthesis of nanocrystalline MFI-zeolite...
Synthesis, structure–activity relationship and molecular docking of 3-oxoaurones and 3-thioaurones as acetylcholinesterase and butyrylcholinesterase inhibitors
Mughal, Ehsan Ullah,Sadiq, Amina,Murtaza, Shahzad,Rafique, Hummera,Zafar, Muhammad Naveed,Riaz, Tauqeer,Khan, Bilal Ahmad,Hameed, Abdul,Khan, Khalid Mohammed
, p. 100 - 106 (2017)
The present study describes efficient an...
Hierarchical mesoporous zeolites: Direct self-assembly synthesis in a conventional surfactant solution by kinetic control over the zeolite seed formation
Zhu, Yan,Hua, Zile,Zhou, Jian,Wang, Lijun,Zhao, Jinjin,Gong, Yun,Wu, Wei,Ruan, Meiling,Shi, Jianlin
, p. 14618 - 14627 (2011)
By kinetic control over the zeolite seed...
Facile one-pot synthesis of flavanones using tetramethylguanidinum-based ionic liquids as catalysts
Zhou, Yan,Huang, Wei,Chen, Xiang-Shu,Song, Zhi-Bin,Tao, Duan-Jian
, p. 1830 - 1836 (2015)
Several tetramethylguanidinum-based ioni...
Synthesis and catalytic activity of amino-functionalized SBA-15 materials with controllable channel lengths and amino loadings
Chen, Shih-Yuan,Huang, Ching-Ya,Yokoi, Toshiyuki,Tang, Chih-Yuan,Huang, Shing-Jong,Lee, Jey-Jau,Chan, Jerry C. C.,Tatsumi, Takashi,Cheng, Soofin
, p. 2233 - 2243 (2012)
Aminopropyl-functionalized SBA-15 mesopo...
Sulfonic acid functionalized mesoporous ZSM-5: Synthesis, characterization and catalytic activity in acidic catalysis
Jin, Hailian,Ansari, Mohd Bismillah,Park, Sang-Eon
, p. 116 - 121 (2015)
Sulfonic acid functionalized mesoporous ...
A new pyrazoline-based fluorescent sensor for Al3+ in aqueous solution
Hu, Shengli,Song, Jingjing,Wu, Gongying,Cheng, Cuixia,Gao, Qing
, p. 1188 - 1194 (2015)
A new pyrazoline-based fluorescent senso...
A simple route to synthesize mesoporous ZSM-5 templated by ammonium-modified chitosan
Jin, Junjiang,Zhang, Xingdi,Li, Yongsheng,Li, Hua,Wu, Wei,Cui, Yunlong,Chen, Qian,Li, Liang,Gu, Jinlou,Zhao, Wenru,Shi, Jianlin
, p. 16549 - 16555 (2012)
Uniform mesoporous zeolite ZSM-5 crystal...
Synthesis and in-silico molecular modelling, DFT studies, antiradical and antihyperglycemic activity of novel vanadyl complexes based on chalcone derivatives
Kaur, Mandeep,Kaushal, Raj
, (2022/01/13)
Diabetes mellitus (DM) will be one of th...
Water stable fluorescent organotin(iv) compounds: aggregation induced emission enhancement and recognition of lead ions in an aqueous system
Capalash, Neena,Kaur, Kulwinder,Kaur, Varinder,Singh, Raghubir
, p. 148 - 161 (2021/12/31)
Herein, synthesis, spectroscopic studies...
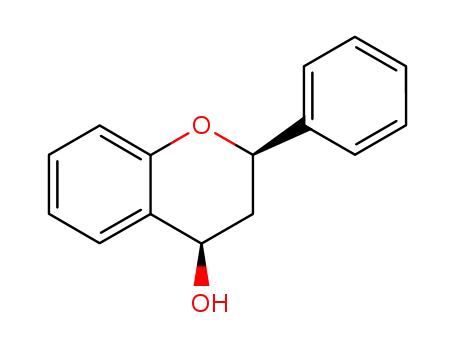
Transfer Hydrogenation of Flavanones and ortho-Hydroxychalcones to 1,3-Diarylpropanols Catalyzed by CNN Pincer Ruthenium Complexes
Soto, Martín,Gotor-Fernández, Vicente,Rodríguez-Solla, Humberto,Baratta, Walter
, p. 2152 - 2157 (2021/03/03)
The transfer hydrogenation of flavanones...
Divergent synthesis of flavones and flavanones from 2′-hydroxydihydrochalconesviapalladium(ii)-catalyzed oxidative cyclization
Son, Seung Hwan,Cho, Yang Yil,Yoo, Hyung-Seok,Lee, Soo Jin,Kim, Young Min,Jang, Hyu Jeong,Kim, Dong Hwan,Shin, Jeong-Won,Kim, Nam-Jung
, p. 14000 - 14006 (2021/04/22)
Divergent and versatile synthetic routes...