

CasNo: 6674-22-2
MF: C9H16N2
Appearance: Colorless to yellow liquid
|
Flammability and Explosibility |
Nonflammable |
|
Synthesis |
The synthesis of DBU is as follows:As a base, sodium hydroxide was used at a concentration of 25% by weight and 2.3 times molar equivalent. A stirrer was placed in a 100 mL screw tube, and 11.5 g (50 mmol) of DBU hydrochloride, 8.5 g of toluene and 18.5 g (116 mmol) of 25% sodium hydroxide aqueous solution were charged at room temperature. It was placed on a magnetic stirrer and mixed for 1 hour with stirring. The mixture was separated with a 100 mL separatory funnel to obtain 18.8 g of the upper layer containing DBU and 18.4 g of the lower layer of the aqueous layer. The upper layer portion was analyzed by gas chromatography and contained 31.9% by weight of DBU, and the yield was 6.0 g (39.4 mmol) in a yield of 78.8%. |
|
Purification Methods |
Fractionally distil DBU under vacuum. Also purify it by chromatography on Kieselgel and eluting with CHCl3/EtOH/25% aqueous NH3 (15:5:2) and checking by IR and MS. [Oediger et al. Chem Ber 99 2012 1962, Angew Chem, Int Ed Engl 6 76 1967, Guggisberg et al. Helv Chim Acta 61 1057 1978, Beilstein 23/5 V 271.] |
|
General Description |
We are committed to bringing you Greener Alternative Products, which adhere to one or more of The 12 Principles of Greener Chemistry. This product has been enhanced for catalytic efficiency. Click here for more information. |
InChI:InChI=1/C9H16N2/c1-2-5-9-10-6-4-8-11(9)7-3-1/h1-8H2/p+1
The article presents a highly effective ...
Thioxanthone-based N-phthalimidoamino ac...
Proton-transfer photopolymerization thro...
The Br?nsted basicities pKaH (i.e., pKa ...
The present invention provides a 1, 8 - ...
PROBLEM TO BE SOLVED: To provide a manuf...
![(2,3,4,6,7,8,9,10-Octahydro-pyrimido[1,2-a]azepin-10-yl)-phenyl-methanol](/upload/2024/7/bfc428bd-90e6-4dde-86d7-95d748eb9b14.png)
(2,3,4,6,7,8,9,10-Octahydro-pyrimido[1,2-a]azepin-10-yl)-phenyl-methanol

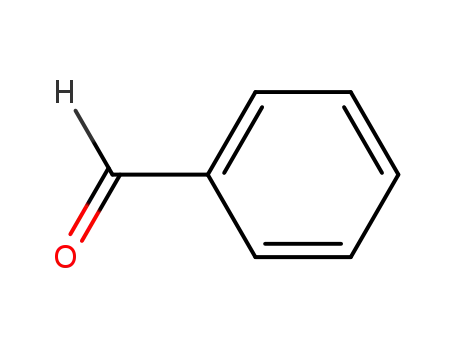
benzaldehyde

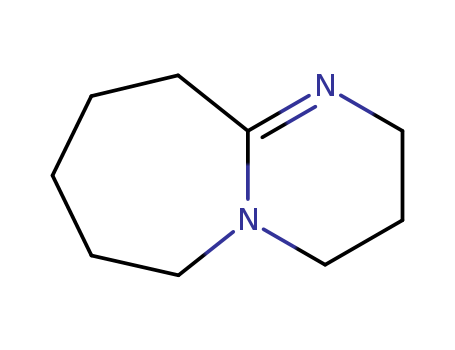
![1,8-diazabicyclo[5.4.0]undec-7-ene](/upload/2024/7/3c729e30-420f-4c35-9813-ab30eec56a5c.png)
1,8-diazabicyclo[5.4.0]undec-7-ene
| Conditions | Yield |
|---|---|
|
at 180 ℃; for 2h; Product distribution;
|
32% |
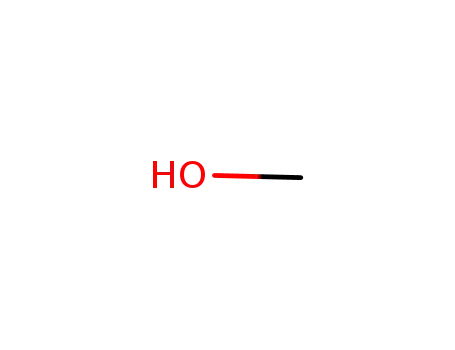
methanol

![methyl 1,8-diazabicyclo[5.4.0]undec-6-ene-8-carboxylate](/upload/2024/7/1855aa9f-3043-450b-a48a-f140a65ff814.png)
methyl 1,8-diazabicyclo[5.4.0]undec-6-ene-8-carboxylate

![1,8-diazabicyclo[5.4.0]undec-7-ene](/upload/2024/7/3c729e30-420f-4c35-9813-ab30eec56a5c.png)
1,8-diazabicyclo[5.4.0]undec-7-ene

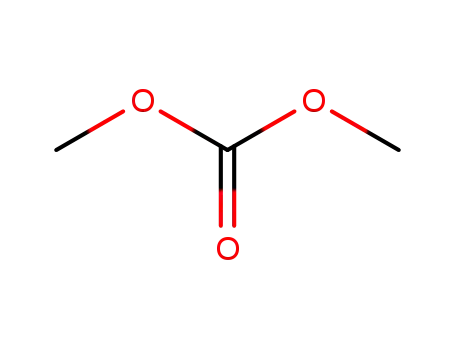
carbonic acid dimethyl ester
| Conditions | Yield |
|---|---|
|
In chloroform-d1; at 19.84 ℃; for 16h; Inert atmosphere;
|
100 %Spectr. 100 %Spectr. |

methanol
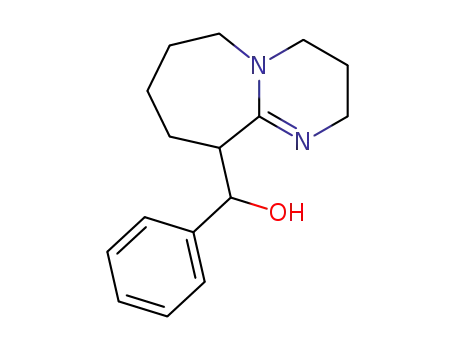
(2,3,4,6,7,8,9,10-Octahydro-pyrimido[1,2-a]azepin-10-yl)-phenyl-methanol
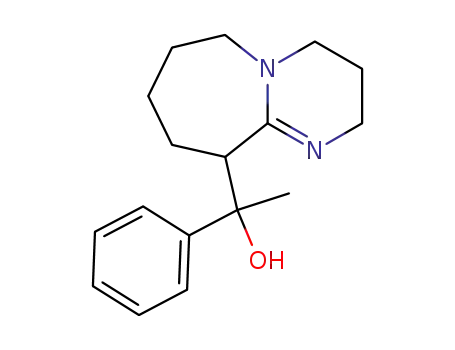
1-(2,3,4,6,7,8,9,10-Octahydro-pyrimido[1,2-a]azepin-10-yl)-1-phenyl-ethanol
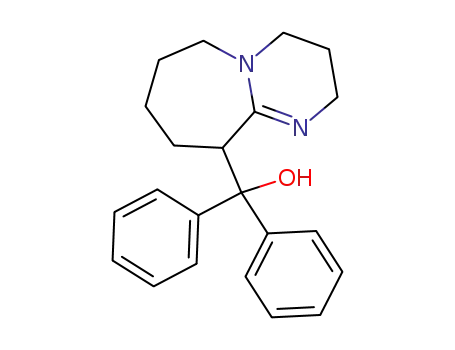
(2,3,4,6,7,8,9,10-Octahydro-pyrimido[1,2-a]azepin-10-yl)-diphenyl-methanol
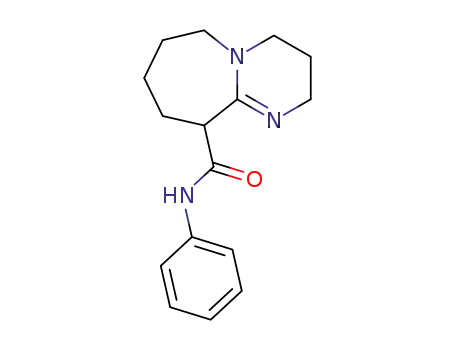
2,3,4,6,7,8,9,10-Octahydro-pyrimido[1,2-a]azepine-10-carboxylic acid phenylamide

(2,3,4,6,7,8,9,10-Octahydro-pyrimido[1,2-a]azepin-10-yl)-diphenyl-methanol
1,3,5-Trinitro-benzene; compound with 2,3,4,6,7,8,9,10-octahydro-pyrimido[1,2-a]azepine